- Home
- CBIC
- Infection Control
- CIC
- CIC - CBIC Certified Infection Control Exam
CBIC CIC CBIC Certified Infection Control Exam Exam Practice Test
CBIC Certified Infection Control Exam Questions and Answers
There has been an outbreak of foodborne illness in the community believed to be associated with attendance at a church festival. Which of the following is the MOST appropriate denominator for calculation of the attack rate?
Options:
People admitted to hospitals with gastrointestinal symptoms
Admission tickets sold to the festival
Dinners served at the festival
Residents in the county who attended the festival
Answer:
DExplanation:
The attack rate, a key epidemiological measure in outbreak investigations, is defined as the proportion of individuals who become ill after exposure to a suspected source, calculated as the number of cases divided by the population at risk. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes accurate outbreak analysis in the "Surveillance and Epidemiologic Investigation" domain, aligning with the Centers for Disease Control and Prevention (CDC) "Principles of Epidemiology in Public Health Practice" (3rd Edition, 2012). The question involves a foodborne illness outbreak linked to a church festival, requiring the selection of the most appropriate denominator to reflect the population at risk.
Option D, "Residents in the county who attended the festival," is the most appropriate denominator. The attack rate should be based on the total number of people exposed to the potential source of the outbreak (i.e., the festival), as this represents the population at risk for developing the foodborne illness. The CDC guidelines for foodborne outbreak investigations recommend using the number of attendees or participants as the denominator when the exposure is tied to a specific event, such as a festival. This approach accounts for all individuals who had the opportunity to consume the implicated food, providing a comprehensive measure of risk. Obtaining an accurate count of attendees may involve festival records, surveys, or estimates, but it directly reflects the exposed population.
Option A, "People admitted to hospitals with gastrointestinal symptoms," is incorrect as a denominator. This represents the number of cases (the numerator), not the total population at risk. Using cases as the denominator would invalidate the attack rate calculation, which requires a distinct population base. Option B, "Admission tickets sold to the festival," could serve as a proxy for attendees if all ticket holders attended, but it may overestimate the at-risk population if some ticket holders did not participate or underestimate it if additional guests attended without tickets. The CDC advises using actual attendance data when available, making this less precise than Option D. Option C, "Dinners served at the festival," is a potential exposure-specific denominator if the illness is linked to a particular meal. However, without confirmation that all cases are tied to a single dinner event (e.g., a specific food item), this is too narrow and may exclude attendees who ate other foods or did not eat but were exposed (e.g., via cross-contamination), making it less appropriate than the broader attendee count.
The CBIC Practice Analysis (2022) and CDC guidelines stress the importance of defining the exposed population accurately for attack rate calculations in foodborne outbreaks. Option D best captures the population at risk associated with festival attendance, making it the most appropriate denominator.
A task force formed to focus on Clostridioides difficile infections (CDIs). The topic of the meeting discussed selecting the correct germicidal wipe. What important factor does the infection preventionist review?
Options:
Cost of a case of wipes
Size of individual wipes
Time the surface remains wet
Correct disposal of the wipe
Answer:
CExplanation:
The correct answer is C, "Time the surface remains wet," as this is the most important factor the infection preventionist (IP) should review when selecting a germicidal wipe for controlling Clostridioides difficile infections (CDIs). According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective environmental cleaning is a critical component of infection prevention, particularly for pathogens like C. difficile, which forms hardy spores that are resistant to many disinfectants. The efficacy of a germicidal wipe depends on the contact time—the duration the surface must remain wet with the disinfectant to ensure the killing of C. difficile spores. This is specified by the manufacturer and supported by guidelines from the Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA), which emphasize that the disinfectant must remain wet on the surface for the full recommended contact time (typically 1-10 minutes for sporicidal agents) to achieve the desired level of disinfection (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols).
Option A (cost of a case of wipes) is a practical consideration for budgeting but is secondary to efficacy in infection control, especially for a high-priority pathogen like C. difficile. Option B (size of individual wipes) may affect coverage and convenience but does not directly impact the wipe’s ability to eliminate the pathogen. Option D (correct disposal of the wipe) is important for preventing cross-contamination and ensuring compliance with waste management protocols, but it is a procedural step after use and not the primary factor in selecting the wipe.
The IP’s review of contact time aligns with CBIC’s focus on evidence-based practices to prevent healthcare-associated infections (HAIs). For C. difficile, which is a leading cause of HAIs, selecting a wipe with an appropriate sporicidal agent and ensuring adequate wet contact time is essential to disrupt transmission, particularly in outbreak settings (CDC Guidelines for Environmental Infection Control in Healthcare Facilities, 2019). This factor directly influences the wipe’s effectiveness, making it the critical review point for the task force.
An infection preventionist receives a call from a healthcare personnel (HCP) who is in the first trimester of pregnancy and has cared for a patient with cytomegalovirus (CMV) pneumonitis. Which of the following actions is MOST appropriate?
Options:
Recommend the pregnant HCP receive immediate post-exposure follow-up.
Reassign the HCP to a low-risk patient care area until second trimester.
Emphasize consistent adherence to Standard Precautions.
Instruct the HCP to wear a mask during patient care activities.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) states that cytomegalovirus (CMV) is a common virus transmitted through direct contact with body fluids, including saliva, urine, blood, and respiratory secretions. In healthcare settings, Standard Precautions are sufficient to prevent CMV transmission, even for pregnant healthcare personnel.
Importantly, routine reassignment, work restriction, or removal from patient care is not recommended for pregnant HCP caring for patients with CMV, including those in the first trimester. The Study Guide emphasizes that the most effective preventive measure is strict adherence to Standard Precautions, particularly hand hygiene and appropriate use of personal protective equipment when contact with body fluids is anticipated. These measures have been shown to significantly reduce the risk of CMV acquisition.
Option A is incorrect because there is no indication for immediate post-exposure evaluation in the absence of a recognized exposure such as a needlestick or mucous membrane contact. Option B is not supported by evidence or guidelines and may contribute to unnecessary workforce restrictions. Option D is insufficient and misleading, as CMV is not transmitted via the airborne route and masking alone does not address the primary transmission risks.
For CIC® exam preparation, it is critical to recognize that education and reinforcement of Standard Precautions—not work exclusion—are the cornerstone of CMV prevention for pregnant healthcare workers.
An infection preventionist is informed that there is a possible cluster of streptococcal meningitis in the neonatal intensive care unit. Which of the following streptococcal serogroops is MOST commonly associated with meningitis in neonates beyond one week of age?
Options:
Group A
Group B
Group C
Group D
Answer:
BExplanation:
Group B Streptococcus (Streptococcus agalactiae) is the most common cause of neonatal bacterial meningitis beyond one week of age.
Step-by-Step Justification:
Group B Streptococcus (GBS) and Neonatal Infections:
GBS is a leading cause of late-onset neonatal meningitis (occurring after 7 days of age).
Infection typically occurs through vertical transmission from the mother or postnatal exposure.
Neonatal Risk Factors:
Premature birth, prolonged rupture of membranes, and maternal GBS colonization increase risk.
Why Other Options Are Incorrect:
A. Group A: Rare in neonates and more commonly associated with pharyngitis and skin infections.
C. Group C: Typically associated with animal infections and rarely affects humans.
D. Group D: Includes Enterococcus, which can cause neonatal infections but is not the most common cause of meningitis.
CBIC Infection Control References:
APIC Text, "Group B Streptococcus and Neonatal Meningitis".
A facility's goal is to increase hand-hygiene compliance from the current 52% to 75% within 12 months. A gap analysis identifies several different issues. Which of the following is BEST suited for summarizing these issues?
Options:
Gantt chart
Flow chart
Ishikawa diagram
Affinity diagram
Answer:
CExplanation:
An Ishikawa diagram (fishbone diagram) is used to visually represent cause-and-effect relationships in problem analysis. It is best for summarizing and categorizing issues found in a gap analysis related to infection prevention.
The APIC Text confirms:
“A fishbone diagram (also called a tree diagram or Ishikawa) allows a team to identify, explore, and graphically display all of the possible causes related to a problem to discover the root cause”.
It’s particularly useful in quality improvement and infection prevention project analysis.
An infection preventionist (IP) is asked to assist in rewriting policies for insertion and maintenance of IV catheters. Which of the following are acceptable for use in site preparation?
Options:
Chloroxylenol or acetone
Povidone-iodine or para-chloro-meta-xylenol (PCMX)
Alcohol or chlorhexidine
Benzalkonium chloride or chlorhexidine
Answer:
CExplanation:
For IV catheter insertion, evidence-based guidance recommends preparing skin with an effective antiseptic agent to reduce skin flora at the insertion site and lower catheter-related infection risk. CDC guidance for prevention of intravascular catheter-related infections specifies that clean skin should be prepared with >0.5% chlorhexidine (CHG) in alcohol for central venous catheter and peripheral arterial catheter insertion and during dressing changes. If CHG is contraindicated, CDC lists tincture of iodine, an iodophor, or 70% alcohol as acceptable alternatives.
Option C (Alcohol or chlorhexidine) is the only answer in which both agents are recognized as appropriate antiseptics for site preparation in intravascular catheter guidance (alcohol as an acceptable antiseptic option; CHG as preferred, typically in alcohol).
The other choices include agents that are not recommended as standard site-prep antiseptics for catheter insertion in major guidelines: acetone is not an antiseptic for vascular access site prep; benzalkonium chloride is generally considered less effective for this purpose compared with CHG/alcohol/iodophors; and PCMX/chloroxylenol is not the typical recommended agent for catheter insertion site antisepsis in these guidelines.
An infection preventionist has been informed that a patient admitted 2 days ago has been diagnosed with chickenpox. Ten employees have had contact with this patient. Those employees with significant exposure may be furloughed after exposure. "Significant exposure" is considered
Options:
greater than one hour of direct patient contact occurring within 24 hours prior to the appearance of lesions.
sharing the same air space for any duration of time after the patient has developed skin lesions.
unprotected contact with respiratory secretions or skin lesions occurring after 12 hours of the appearance of lesions.
irrelevant unless the employee has a negative varicella antibody titer.
Answer:
BExplanation:
Chickenpox (varicella) is primarily spread through airborne transmission, and exposure is defined by being in the same airspace with a contagious person (from 1-2 days before rash onset until lesions are crusted), even if briefly.
The APIC Text states:
“Significant exposure is defined as being in the same room or airspace during the period of infectivity, regardless of duration”.
This reflects airborne precaution definitions and CDC exposure management guidelines for varicella.
Occupational Health contacts the Infection Preventionist (IP) regarding exposure of a patient to an employee's blood during surgery. The employee is negative for bloodborne pathogens. What is the NEXT step regarding informing the patient of the exposure?
Options:
Disclose the exposure to the patient's surgeon and allow surgeon to determine if patient should be informed
Disclose the exposure to the patient with the information that the staff member is negative for all bloodborne pathogens
Since this was a solid needle and not a hollow bore needed, follow up is not required or need to be disclosed
The patient does not need to be informed since the employee is negative for all bloodborne pathogens
Answer:
BExplanation:
Even if the healthcare worker is negative for bloodborne pathogens, the patient has the right to be informed of a potential exposure. Transparency builds trust and aligns with ethical obligations in patient care.
The APIC Text states:
“Providers should inform patients when an HAI or other exposure event occurs, regardless of whether the exposure results in harm or is caused by negligence.” Courts and professional guidelines support disclosure.
CBIC and OSHA guidelines emphasize prompt and transparent reporting of exposures.
Options C and D are incorrect because the lack of infection does not negate the ethical duty to inform the patient.
The infection preventionist recognizes that construction barriers are a key component of the Infection Control Risk Assessment (ICRA). The MOST important factor to consider is that construction barriers should:
Options:
Be constructed to withstand normal heating, ventilation, and air conditioning (HVAC) airflow rates.
Provide sealed covers for air intakes and exhausts.
Be able to contain dust or infectious microorganisms generated by the project.
Have walk-off mats that are changed daily.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that the primary purpose of construction barriers within an Infection Control Risk Assessment (ICRA) is to prevent the dissemination of dust and potentially infectious microorganisms generated during construction, renovation, or maintenance activities. Construction activities can aerosolize fungal spores (such as Aspergillus), bacteria, and other particulate matter that pose a significant risk to immunocompromised patients and other vulnerable populations.
Barriers must therefore be designed and maintained to effectively contain dust and microorganisms at the source, preventing their migration into occupied patient care areas. This containment function is the cornerstone of infection prevention during construction and directly aligns with ICRA goals of risk reduction and patient safety.
While the other options describe supportive or secondary considerations, they are not the most critical factor. Withstanding HVAC airflow (Option A) is important, but it serves the larger goal of containment. Sealing air intakes and exhausts (Option B) is a specific engineering control that may be used as part of containment strategies but does not define the primary purpose of barriers. Walk-off mats (Option D) are useful adjunctive controls but are insufficient alone to prevent airborne transmission of contaminants.
For CIC® exam preparation, it is essential to recognize that containment of dust and infectious agents is the defining function of construction barriers within an ICRA, and all other measures support this central objective.
==========
Which water type is suitable for drinking yet may still be a risk for disease transmission?
Options:
Purified water
Grey water
Potable water
Distilled water
Answer:
CExplanation:
To determine which water type is suitable for drinking yet may still pose a risk for disease transmission, we need to evaluate each option based on its definition, treatment process, and potential for contamination, aligning with infection control principles as outlined by the Certification Board of Infection Control and Epidemiology (CBIC).
A. Purified water: Purified water undergoes a rigorous treatment process (e.g., reverse osmosis, distillation, or deionization) to remove impurities, contaminants, and microorganisms. This results in water that is generally safe for drinking and has a very low risk of disease transmission when properly handled and stored. However, if the purification process is compromised or if contamination occurs post-purification (e.g., due to improper storage or distribution), there could be a theoretical risk. Nonetheless, purified water is not typically considered a primary source of disease transmission under standard conditions.
B. Grey water: Grey water refers to wastewater generated from domestic activities such as washing dishes, laundry, or bathing, which may contain soap, food particles, and small amounts of organic matter. It is not suitable for drinking due to its potential contamination with pathogens (e.g., bacteria, viruses) and chemicals. Grey water is explicitly excluded from potable water standards and poses a significant risk for disease transmission, making it an unsuitable choice for this question.
C. Potable water: Potable water is water that meets regulatory standards for human consumption, as defined by organizations like the World Health Organization (WHO) or the U.S. Environmental Protection Agency (EPA). It is treated to remove harmful pathogens and contaminants, making it safe for drinking under normal circumstances. However, despite treatment, potable water can still pose a risk for disease transmission if the distribution system is contaminated (e.g., through biofilms, cross-connections, or inadequate maintenance of pipes). Outbreaks of waterborne diseases like Legionnaires' disease or gastrointestinal infections have been linked to potable water systems, especially in healthcare settings. This makes potable water the best answer, as it is suitable for drinking yet can still carry a risk under certain conditions.
D. Distilled water: Distilled water is produced by boiling water and condensing the steam, which removes most impurities, minerals, and microorganisms. It is highly pure and safe for drinking, often used in medical and laboratory settings. Similar to purified water, the risk of disease transmission is extremely low unless contamination occurs after distillation due to improper handling or storage. Like purified water, it is not typically associated with disease transmission risks in standard use.
The key to this question lies in identifying a water type that is both suitable for drinking and has a documented potential for disease transmission. Potable water fits this criterion because, while it is intended for consumption and meets safety standards, it can still be a vector for disease if the water supply or distribution system is compromised. This is particularly relevant in infection control, where maintaining water safety in healthcare facilities is a critical concern addressed by CBIC guidelines.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain III: Prevention and Control of Infectious Diseases, which highlights the importance of water safety and the risks of contamination in potable water systems.
CBIC Examination Content Outline, Domain IV: Environment of Care, which includes managing waterborne pathogens (e.g., Legionella) in potable water supplies.
A patient with pertussis can be removed from Droplet Precautions after
Options:
direct fluorescent antibody and/or culture are negative.
five days of appropriate antibiotic therapy.
the patient has been given pertussis vaccine.
the paroxysmal stage has ended.
Answer:
BExplanation:
A patient with pertussis (whooping cough) should remain on Droplet Precautions to prevent transmission. According to APIC guidelines, patients with pertussis can be removed from Droplet Precautions after completing at least five days of appropriate antimicrobial therapy and showing clinical improvement.
Why the Other Options Are Incorrect?
A. Direct fluorescent antibody and/or culture are negative – Laboratory results may not always detect pertussis early, and false negatives can occur.
C. The patient has been given pertussis vaccine – The vaccine prevents but does not treat pertussis, and it does not shorten the period of contagiousness.
D. The paroxysmal stage has ended – The paroxysmal stage (severe coughing fits) can last weeks, but infectiousness decreases with antibiotics.
CBIC Infection Control Reference
According to APIC guidelines, Droplet Precautions should continue until the patient has received at least five days of antimicrobial therapy.
An infection preventionist is preparing a report about an outbreak of scabies in a long-term care facility. How would this information be displayed in an epidemic curve?
Options:
List case names, room numbers, and date the infestation was identified using a logarithmic scale.
List case medical record numbers and the number of days in the facility to date of onset, showing data in a scatter plot.
Prepare a bar graph with no patient identifiers showing the number of cases over a specific period of time.
Prepare a scatter plot by patient location showing case prevalence over a specific period of time.
Answer:
CExplanation:
An epidemic curve, commonly used in infection prevention and control to visualize the progression of an outbreak, is a graphical representation of the number of cases over time. According to the principles outlined by the Certification Board of Infection Control and Epidemiology (CBIC), an epidemic curve is most effectively displayed using a bar graph or histogram that tracks the number of new cases by date or time interval (e.g., daily, weekly) without revealing patient identifiers, ensuring compliance with privacy regulations such as HIPAA. Option C aligns with this standard practice, as it specifies preparing a bar graph with no patient identifiers, focusing solely on the number of cases over a specific period. This allows infection preventionists to identify patterns, such as the peak of the outbreak or potential sources of transmission, while maintaining confidentiality.
Option A is incorrect because listing case names and room numbers with a logarithmic scale violates patient privacy and is not a standard method for constructing an epidemic curve. Logarithmic scales are typically used for data with a wide range of values, but they are not the preferred format for epidemic curves, which prioritize clarity over time. Option B is also incorrect, as using medical record numbers and scatter plots to show days in the facility to onset does not align with the definition of an epidemic curve, which focuses on case counts over time rather than individual patient timelines or scatter plot formats. Option D is inappropriate because a scatter plot by patient location emphasizes spatial distribution rather than the temporal progression central to an epidemic curve. While location data can be useful in outbreak investigations, it is typically analyzed separately from the epidemic curve.
The CBIC emphasizes the importance of epidemic curves in the "Identification of Infectious Disease Processes" domain, where infection preventionists use such tools to monitor and control outbreaks (CBIC Practice Analysis, 2022). Specifically, the use of anonymized data in graphical formats is a best practice to protect patient information while providing actionable insights, as detailed in the CBIC Infection Prevention and Control (IPC) guidelines.
An infection preventionist (IP) reviews research from a current publication and compares the data to the IP’s facility data. This is an example of:
Options:
Benchmarking
Data collection
Linear regression
Data mining
Answer:
AExplanation:
The Certification Study Guide (6th edition) defines benchmarking as the process of comparing an organization’s performance data with external reference points, such as published research, national databases, or peer institutions. In this scenario, the infection preventionist is comparing the facility’s data to findings from a current publication, which clearly represents benchmarking activity.
Benchmarking allows infection preventionists to determine how their facility is performing relative to recognized standards, evidence-based outcomes, or peer performance. The study guide emphasizes that benchmarking is essential for identifying performance gaps, prioritizing improvement initiatives, and supporting data-driven decision-making. It is frequently used when evaluating infection rates, compliance metrics, and outcomes associated with prevention strategies.
The other options do not accurately describe this activity. Data collection refers to the gathering of raw data, not comparison. Linear regression is a statistical analysis method used to examine relationships between variables over time and is not implied in this scenario. Data mining involves exploring large datasets to identify patterns or associations, typically without a predefined comparison target.
CIC exam questions often test understanding of data use versus data analysis methods. Recognizing benchmarking as the comparison of internal performance to external standards is a foundational competency for infection preventionists. This practice supports quality improvement, regulatory compliance, and leadership reporting.
Major construction and renovations are planned for a hospital’s operating suite, and a meeting is scheduled to plan for construction activities. Aside from the infection preventionist, and representatives from environmental services and engineering, who else should be included in these planning conversations?
Options:
Operating room nurse manager
Chief operating officer
Plumbing supervisor
Director of public relations
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that multidisciplinary collaboration is essential when planning construction or renovation projects in patient care areas, especially high-risk locations such as operating suites. In addition to infection prevention, environmental services, and engineering, the operating room nurse manager must be actively involved in construction planning discussions.
The operating room nurse manager represents frontline clinical operations and has direct knowledge of surgical workflows, patient movement, sterile processing needs, case scheduling, and staff practices. Their involvement ensures that construction activities are coordinated to minimize disruption to patient care, maintain sterile environments, and reduce infection risks associated with dust, airflow changes, and traffic patterns. The nurse manager also plays a key role in communicating construction-related precautions and practice changes to surgical staff.
While senior leadership (Option B) may provide oversight, they are not typically involved in detailed infection control planning. The plumbing supervisor (Option C) may be consulted for specific infrastructure issues but does not represent clinical operations. The director of public relations (Option D) is not relevant to construction-related infection risk planning.
The Study Guide highlights that ICRA planning must include clinical leadership from affected areas to ensure that infection prevention measures are practical, effective, and consistently implemented. Including the operating room nurse manager is therefore essential for safe construction planning and is a frequently tested CIC® exam concept.
==========
The infection preventionist (IP) collaborates with the Intravenous Therapy team to select the best antiseptic for use during the insertion of an intravascular device for adults. For a patient with no contraindications, what antiseptic should the IP suggest?
Options:
Chlorhexidine
Povidone-iodine
Alcohol
Antibiotic ointment
Answer:
AExplanation:
The selection of an appropriate antiseptic for the insertion of an intravascular device (e.g., peripheral or central venous catheters) is a critical infection prevention measure to reduce the risk of catheter-related bloodstream infections (CRBSIs). The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes evidence-based practices in the "Prevention and Control of Infectious Diseases" domain, which includes adhering to guidelines for aseptic technique during invasive procedures. The Centers for Disease Control and Prevention (CDC) provides specific recommendations for skin antisepsis, as outlined in the "Guidelines for the Prevention of Intravascular Catheter-Related Infections" (2017).
Option A, chlorhexidine, is the preferred antiseptic for skin preparation prior to intravascular device insertion in adults with no contraindications. Chlorhexidine, particularly in a 2% chlorhexidine gluconate (CHG) with 70% isopropyl alcohol solution, is recommended by the CDC due to its broad-spectrum antimicrobial activity, residual effect (which continues to kill bacteria after application), and superior efficacy compared to other agents in reducing CRBSI rates. Studies cited in the CDC guidelines demonstrate that chlorhexidine-based preparations significantly lower infection rates compared to povidone-iodine or alcohol alone, making it the gold standard for this procedure when tolerated by the patient.
Option B, povidone-iodine, is an alternative antiseptic that can be used for skin preparation. It is effective against a wide range of microorganisms and is often used when chlorhexidine is contraindicated (e.g., in patients with chlorhexidine allergy). However, its efficacy is less persistent than chlorhexidine, and it requires longer drying time, which can be a limitation in busy clinical settings. The CDC considers povidone-iodine a second-line option unless chlorhexidine is unavailable or unsuitable. Option C, alcohol (e.g., 70% isopropyl or ethyl alcohol), has rapid bactericidal activity but lacks a residual effect, making it less effective for prolonged protection during catheter dwell time. It is often used as a component of chlorhexidine-alcohol combinations but is not recommended as a standalone antiseptic for intravascular device insertion. Option D, antibiotic ointment, is not appropriate for skin preparation during insertion. Antibiotic ointments (e.g., bacitracin or mupirocin) are sometimes applied to catheter sites post-insertion to prevent infection, but their use is discouraged by the CDC due to the risk of promoting antibiotic resistance and fungal infections, and they are not classified as antiseptics for initial skin antisepsis.
The CBIC Practice Analysis (2022) supports the adoption of CDC-recommended practices, and the 2017 CDC guidelines explicitly state that chlorhexidine-based preparations with alcohol should be used for skin antisepsis unless contraindicated. For a patient with no contraindications, the infection preventionist should suggest chlorhexidine to optimize patient safety and align with best practices.
A healthcare professional in a clinical microbiology laboratory is concerned about routine exposure to Neisseria meningitidis in culture. The healthcare professional last received the Meningococcal vaccine 8 years ago. What recommendation should be given to the healthcare professional regarding their meningococcal vaccination?
Options:
They are due for a booster as it has been over 5 years.
They are due for a booster as it has been over 7 years.
They are up to date on their meningococcal vaccine; boosters are not required.
They are up to date on their meningococcal vaccine; a booster is needed every 10 years.
Answer:
BExplanation:
The correct answer is B, "They are due for a booster as it has been over 7 years," as this is the appropriate recommendation for the healthcare professional regarding their meningococcal vaccination. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), healthcare professionals with routine exposure to Neisseria meningitidis, such as those in clinical microbiology laboratories, are at increased risk of meningococcal disease due to potential aerosol or droplet exposure during culture handling. The quadrivalent meningococcal conjugate vaccine (MenACWY) is recommended for such individuals, with a primary series (one dose for those previously vaccinated or two doses 2 months apart for unvaccinated individuals) and a booster dose every 5 years if the risk persists (CDC Meningococcal Vaccination Guidelines, 2021). However, for laboratory workers with ongoing exposure, the ACIP specifies a booster interval of every 5 years from the last dose, but this is often interpreted in practice as aligning with the 5-7 year range depending on risk assessment and institutional policy. Since the healthcare professional received the vaccine 8 years ago and works in a high-risk setting, a booster is due, with the 7-year threshold being a practical midpoint for this scenario (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents).
Option A (they are due for a booster as it has been over 5 years) is close but slightly premature based on the 8-year interval, though it reflects the general 5-year booster guideline for high-risk groups; the 7-year option better matches the specific timeframe. Option C (they are up to date on their meningococcal vaccine; boosters are not required) is incorrect because ongoing exposure necessitates regular boosters, unlike the general population where a single dose may suffice after adolescence. Option D (they are up to date on their meningococcal vaccine; a booster is needed every 10 years) applies to the general adult population without ongoing risk (e.g., post-adolescence vaccination), not to laboratory workers with continuous exposure, where the interval is shorter.
The recommendation for a booster aligns with CBIC’s emphasis on protecting healthcare personnel from occupational exposure to communicable diseases, ensuring compliance with evidence-based immunization practices (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This supports the prevention of meningococcal disease outbreaks in healthcare settings.
In a retrospective case-control study, the initial case group is composed of persons
Options:
with the disease
without the disease.
with the risk factor under investigation
without the risk factor under investigation
Answer:
AExplanation:
In a retrospective case-control study, cases and controls are selected based on disease status. The case group is composed of individuals who have the disease (cases), while the control group consists of individuals without the disease. This design allows researchers to look back in time to assess exposure to potential risk factors.
Step-by-Step Justification:
Selection of Cases and Controls:
Cases: Individuals who already have the disease.
Controls: Individuals without the disease but similar in other aspects.
Direction of Study:
A retrospective study moves backward from the disease outcome to investigate potential causes or risk factors.
Data Collection:
Uses past medical records, interviews, and laboratory results to determine past exposures.
Common Use:
Useful for studying rare diseases since cases have already occurred, making it cost-effective compared to cohort studies.
Why Other Options Are Incorrect:
B. without the disease: (Incorrect) This describes the control group, not the case group.
C. with the risk factor under investigation: (Incorrect) Risk factors are identified after selecting cases and controls.
D. without the risk factor under investigation: (Incorrect) The study investigates whether cases had prior exposure, not whether they lacked a risk factor.
CBIC Infection Control References:
APIC Text, Chapter on Epidemiologic Study Design.
An infection preventionist has been asked to participate in a process improvement team to standardize disinfection and sterilization practices. Team activities should include all of the following EXCEPT:
Options:
Observing disinfection and sterilization practices.
Asking central supply and operating room managers to join the team.
Performing a literature review on central supply and sterilization.
Conducting outcome measurement after all changes are implemented.
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that effective process improvement relies on a structured, data-driven approach that includes baseline assessment, intervention, and ongoing evaluation. A key principle of quality improvement is that outcomes must be measured before and after changes are implemented in order to determine whether an intervention resulted in improvement.
Option D is the correct “EXCEPT” choice because limiting outcome measurement to only after changes are implemented prevents meaningful comparison and makes it impossible to determine effectiveness. Without baseline data, improvements cannot be quantified, trends cannot be assessed, and unintended consequences may go unrecognized. The Study Guide stresses that baseline measurements are essential to evaluate process performance and to support evidence-based decision-making.
Options A, B, and C are all appropriate and expected activities. Direct observation helps identify workflow gaps and variation in practice. Inclusion of central supply and operating room leadership ensures multidisciplinary engagement and operational insight. Conducting a literature review supports alignment with current evidence, standards, and best practices for disinfection and sterilization.
For the CIC® exam, it is important to recognize that continuous measurement throughout the improvement cycle—not only after implementation—is required for successful standardization and sustainability of infection prevention practices.
When evaluating environmental cleaning and disinfectant products as a part of the product evaluation committee, which of the following is responsible for providing information regarding clinical trials?
Options:
Infection Preventionist
Clinical representatives
Environmental Services
Manufacturer representatives
Answer:
DExplanation:
The correct answer is D, "Manufacturer representatives," as they are responsible for providing information regarding clinical trials when evaluating environmental cleaning and disinfectant products as part of the product evaluation committee. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, manufacturers are the primary source of data on the efficacy, safety, and performance of their products, including clinical trial results that demonstrate the disinfectant’s ability to reduce microbial load or prevent healthcare-associated infections (HAIs) (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This information is critical for the committee to assess whether the product meets regulatory standards (e.g., EPA registration) and aligns with infection prevention goals, and it is typically supported by documentation such as peer-reviewed studies or trial data provided by the manufacturer.
Option A (Infection Preventionist) plays a key role in evaluating the product’s fit within infection control practices and may contribute expertise or conduct internal assessments, but they are not responsible for providing clinical trial data, which originates from the manufacturer. Option B (Clinical representatives) can offer insights into clinical usage and outcomes but rely on manufacturer data for trial evidence rather than generating it. Option C (Environmental Services) focuses on the practical application and cleaning processes but lacks the authority or resources to conduct or provide clinical trial information.
The reliance on manufacturer representatives aligns with CBIC’s emphasis on evidence-based decision-making in product selection, ensuring that the product evaluation committee bases its choices on robust, manufacturer-supplied clinical data (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies). This approach supports the safe and effective implementation of environmental cleaning products in healthcare settings.
A change in the disinfection protocol is indicated for which of the following scenarios?
Options:
A high-level disinfectant being used for diaphragm fitting rings
Sodium hypochlorite being used for blood pressure cuffs
An enzymatic solution being used for rectal probes
2% glutaraldehyde being used for cryosurgical probes
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of applying Spaulding’s classification to determine appropriate cleaning, disinfection, and sterilization levels for medical devices based on their intended use. According to this framework, rectal probes are classified as semi-critical devices because they come into contact with mucous membranes. Semi-critical devices require at least high-level disinfection after thorough cleaning.
An enzymatic solution, as listed in option C, is not a disinfectant. Enzymatic detergents are designed solely for cleaning, meaning they help remove organic material such as blood, mucus, and feces, but they do not kill microorganisms. Using an enzymatic solution alone for rectal probes is therefore inadequate and represents an improper disinfection practice, making this the scenario that clearly requires a protocol change.
Option A is acceptable because diaphragm fitting rings are noncritical devices that contact intact skin and may be safely processed using high-level disinfection. Option B is appropriate because blood pressure cuffs are noncritical items and can be disinfected using low- to intermediate-level disinfectants such as sodium hypochlorite. Option D is also appropriate, as cryosurgical probes are semi-critical devices and 2% glutaraldehyde is an accepted high-level disinfectant.
Recognizing the distinction between cleaning versus disinfection and applying the correct level of processing is a core competency for infection preventionists and a frequently tested concept on the CIC® exam.
==========
How can infection preventionists BEST educate on a new infection prevention protocol for adoption across different departments within an organization?
Options:
Relate relevance of each department’s role using scenarios.
Utilize a passive learning method to accommodate for diversity.
Assign reading and review of protocol with manager sign-off.
Tie protocol compliance metric to annual performance review.
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that effective education for adult learners is most successful when it is relevant, interactive, and role-specific. Relating the new infection prevention protocol to each department’s responsibilities using realistic scenarios is the most effective educational strategy for organization-wide adoption.
Scenario-based education is an active learning method, which engages participants in problem-solving and application of knowledge rather than passive receipt of information. By tailoring scenarios to departmental workflows—such as nursing, environmental services, laboratory, or ancillary departments—staff can clearly understand how the protocol affects their daily practice and how their actions contribute to infection prevention outcomes. This approach improves comprehension, retention, and compliance.
Option B is incorrect because passive learning methods (e.g., lectures or handouts alone) are less effective for behavior change and adult learning. Option C relies on administrative acknowledgment rather than understanding and does not ensure competency or consistent application. Option D may support accountability but does not educate staff or build understanding during initial implementation.
The Study Guide stresses that infection preventionists must act as educators and change agents, adapting teaching strategies to diverse audiences. Using scenario-based, department-specific education aligns with adult learning principles, promotes engagement, and facilitates sustainable practice change—making it the best approach and a key concept for the CIC® exam.
==========
The infection preventionist (IP) is reviewing a laboratory report that indicates the presence of Enterococcus faecium in a 76-year-old patient’s urine culture. The patient has no symptoms of a urinary tract infection. The IP’s accurate interpretation of this result is that the patient:
Options:
Should be placed in isolation due to the risk of airborne transmission.
Has an active infection and requires immediate treatment with antibiotics.
Is colonized with the bacteria and does not require treatment at this time.
Has a pseudo-infection, which could be caused by contamination of the sample.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of distinguishing colonization from infection when interpreting microbiology results. Colonization refers to the presence of microorganisms on or within the body without causing clinical signs or symptoms of disease. In older adults, especially those in healthcare settings, asymptomatic bacteriuria is common and does not meet criteria for a urinary tract infection (UTI).
In this scenario, the presence of Enterococcus faecium in a urine culture in the absence of urinary symptoms—such as dysuria, urgency, fever, or suprapubic pain—indicates colonization rather than infection. The Study Guide notes that treating asymptomatic bacteriuria does not improve patient outcomes and may contribute to antimicrobial resistance, adverse drug events, and unnecessary healthcare costs. Therefore, antibiotics are not indicated.
Option A is incorrect because Enterococcus species are not transmitted via the airborne route; Standard Precautions are sufficient. Option B is incorrect because laboratory findings alone do not define infection without corresponding clinical symptoms. Option D is less accurate because contamination is more likely with mixed flora or improper collection; isolation of a known urinary colonizer in an asymptomatic patient is more consistent with colonization.
Accurate interpretation of such findings supports antimicrobial stewardship principles and aligns with evidence-based infection prevention practices tested on the CIC® exam.
==========
The infection preventionist recognizes that facility renovation projects are associated with risk for healthcare-associated infections (HAIs). What is the primary rationale for this risk?
Options:
Increased numbers of construction staff resulting in congested workspaces
Improper training regarding hand hygiene for contracted workers
Trash removal using uncovered carts
Environmental dispersal of microorganisms
Answer:
DExplanation:
The Certification Study Guide (6th edition) identifies environmental dispersal of microorganisms as the primary reason healthcare construction and renovation activities increase the risk of healthcare-associated infections (HAIs). Construction activities such as demolition, drilling, and ceiling penetration disturb dust and building materials that may harbor fungal spores and other microorganisms, particularly Aspergillus species. Once aerosolized, these organisms can spread through airflow to patient care areas if containment and ventilation controls are inadequate.
The study guide emphasizes that immunocompromised patients—such as those in oncology units, transplant units, and intensive care settings—are especially vulnerable to infections caused by airborne fungi released during construction. Numerous outbreaks of invasive aspergillosis have been linked directly to renovation and construction projects that lacked appropriate infection control risk assessment (ICRA) measures.
The incorrect options describe secondary or contributory issues but are not the primary mechanism of infection risk. While increased personnel traffic, hand hygiene training, and waste handling are important considerations, they do not represent the central hazard posed by construction. The fundamental risk is airborne dissemination of microorganisms from disrupted environmental reservoirs.
CIC exam questions frequently test knowledge of construction-related infection risks and the importance of engineering controls such as negative pressure containment, HEPA filtration, and dust barriers. Recognizing environmental dispersal as the primary risk underscores why rigorous planning and infection control oversight are essential during renovation projects.
The infection preventionist observed a caregiver entering a room without performing hand hygiene. The BEST response would be to
Options:
post additional signage to remind caregivers to wash before entry.
provide immediate feedback and education to the caregiver.
install hand hygiene dispensers in more convenient areas.
design a unit-based education program.
Answer:
BExplanation:
Immediate feedback is a best practice in behavior correction and performance improvement. In hand hygiene non-compliance, real-time intervention allows for immediate correction, education, and reinforcement of infection prevention policies.
The APIC/JCR Workbook recommends:
“Provide simulation training… that provides immediate feedback—for example, how to properly insert a urinary catheter or perform hand hygiene.” This supports behavior change and staff learning.
The APIC Text emphasizes that real-time, direct feedback is more effective than passive measures like signage or delayed education campaigns.
A nurse claims to have acquired hepatitis A virus infection as the result of occupational exposure. The source patient had an admitting diagnosis of viral hepatitis. Further investigation of this incident reveals a 5-day interval between exposure and onset of symptoms in the nurse. The patient has immunoglobulin G antibodies to hepatitis A. From the evidence, the infection preventionist may correctly conclude which of the following?
Options:
The nurse should be given hepatitis A virus immunoglobulin.
The evidence at this time fails to support the nurse's claim.
The patient has serologic evidence of recent hepatitis A viral infection.
The 5-day incubation period is consistent with hepatitis A virus transmission.
Answer:
BExplanation:
The infection preventionist’s (IP) best conclusion, based on the provided evidence, is that the evidence at this time fails to support the nurse's claim of acquiring hepatitis A virus (HAV) infection through occupational exposure. This conclusion is grounded in the clinical and epidemiological understanding of HAV, as aligned with the Certification Board of Infection Control and Epidemiology (CBIC) guidelines. Hepatitis A typically has an incubation period ranging from 15 to 50 days, with an average of approximately 28-30 days, following exposure to the virus (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.3 - Apply principles of epidemiology). The reported 5-day interval between exposure and symptom onset in the nurse is significantly shorter than the expected incubation period, making it inconsistent with HAV transmission. Additionally, the presence of immunoglobulin G (IgG) antibodies in the source patient indicates past exposure or immunity to HAV, rather than an active or recent infection, which would typically be associated with immunoglobulin M (IgM) antibodies during the acute phase.
Option A (the nurse should be given hepatitis A virus immunoglobulin) is not supported because post-exposure prophylaxis with HAV immunoglobulin is recommended only within 14 days of exposure to a confirmed case with active infection, and the evidence here does not confirm a recent exposure or active case. Option C (the patient has serologic evidence of recent hepatitis A viral infection) is incorrect because IgG antibodies signify past infection or immunity, not a recent infection, which would require IgM antibodies. Option D (the 5-day incubation period is consistent with hepatitis A virus transmission) is inaccurate due to the mismatch with the known incubation period of HAV.
The IP’s role includes critically evaluating epidemiological data to determine the likelihood of transmission events. The discrepancy in the incubation period and the serologic status of the patient suggest that the nurse’s claim may not be substantiated by the current evidence, necessitating further investigation rather than immediate intervention or acceptance of the claim. This aligns with CBIC’s emphasis on accurate identification and investigation of infectious disease processes (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.2 - Investigate suspected outbreaks or exposures).
A patient with suspected active tuberculosis is being transferred from a mental health facility to a medical center by emergency medical services. Which of the following should an infection preventionist recommend to the emergency medical technician (EMT)?
Options:
Place a surgical mask on both the patient and the EMT.
Place an N95 respirator on both the patient and the EMT.
Place an N95 respirator on the patient and a surgical mask on the EMT.
Place a surgical mask on the patient and an N95 respirator on the EMT.
Answer:
CExplanation:
Active tuberculosis (TB) is an airborne disease transmitted through the inhalation of droplet nuclei containing Mycobacterium tuberculosis. Effective infection control measures are critical during patient transport to protect healthcare workers, such as emergency medical technicians (EMTs), and to prevent community spread. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the use of appropriate personal protective equipment (PPE) and source control as key strategies in the "Prevention and Control of Infectious Diseases" domain, aligning with guidelines from the Centers for Disease Control and Prevention (CDC).
For a patient with suspected active TB, the primary goal is to contain the infectious particles at the source (the patient) while ensuring the EMT is protected from inhalation exposure. Option C, placing an N95 respirator on the patient and a surgical mask on the EMT, is the most appropriate recommendation. The N95 respirator on the patient serves as source control by filtering the exhaled air, reducing the dispersion of infectious droplets. However, fitting an N95 respirator on the patient may be challenging, especially in an emergency setting or if the patient is uncooperative, so a surgical mask is often used as an alternative source control measure. For the EMT, a surgical mask provides a basic barrier but does not offer the same level of respiratory protection as an N95 respirator. The CDC recommends that healthcare workers, including EMTs, use an N95 respirator (or higher-level respiratory protection) when in close contact with a patient with suspected or confirmed active TB, unless an airborne infection isolation room is available, which is not feasible during transport.
Option A is incorrect because placing a surgical mask on both the patient and the EMT does not provide adequate respiratory protection for the EMT. Surgical masks are not designed to filter small airborne particles like those containing TB bacilli and do not meet the N95 standard required for airborne precautions. Option B is impractical and unnecessary, as placing an N95 respirator on both the patient and the EMT is overly restrictive and logistically challenging, especially for the patient during transport. Option D reverses the PPE roles, placing the surgical mask on the patient (insufficient for source control) and the N95 respirator on the EMT (appropriate for protection but misaligned with the need to control the patient’s exhalation). The CBIC and CDC guidelines prioritize source control on the patient and respiratory protection for the healthcare worker, making Option C the best fit.
This recommendation is consistent with the CBIC’s emphasis on implementing transmission-based precautions (CDC, 2005, Guideline for Preventing the Transmission of Mycobacterium tuberculosis in Healthcare Settings) and the use of PPE tailored to the mode of transmission, as outlined in the CBIC Practice Analysis (2022).
When conducting a literature search which of the following study designs may provide the best evidence of a direct causal relationship between the experimental factor and the outcome?
Options:
A case report
A descriptive study
A case control study
A randomized-controlled trial
Answer:
DExplanation:
To determine the best study design for providing evidence of a direct causal relationship between an experimental factor and an outcome, it is essential to understand the strengths and limitations of each study design listed. The goal is to identify a design that minimizes bias, controls for confounding variables, and establishes a clear cause-and-effect relationship.
A. A case report: A case report is a detailed description of a single patient or a small group of patients with a particular condition or outcome, often including the experimental factor of interest. While case reports can generate hypotheses and highlight rare occurrences, they lack a control group and are highly susceptible to bias. They do not provide evidence of causality because they are observational and anecdotal in nature. This makes them the weakest design for establishing a direct causal relationship.
B. A descriptive study: Descriptive studies, such as cross-sectional or cohort studies, describe the characteristics or outcomes of a population without manipulating variables. These studies can identify associations between an experimental factor and an outcome, but they do not establish causality due to the absence of randomization or control over confounding variables. For example, a descriptive study might show that a certain infection rate is higher in a group exposed to a specific factor, but it cannot prove the factor caused the infection without further evidence.
C. A case control study: A case control study compares individuals with a specific outcome (cases) to those without (controls) to identify factors that may contribute to the outcome. This retrospective design is useful for studying rare diseases or outcomes and can suggest associations. However, it is prone to recall bias and confounding, and it cannot definitively prove causation because the exposure is not controlled or randomized. It is stronger than case reports or descriptive studies but still falls short of establishing direct causality.
D. A randomized-controlled trial (RCT): An RCT is considered the gold standard for establishing causality in medical and scientific research. In an RCT, participants are randomly assigned to either an experimental group (exposed to the factor) or a control group (not exposed or given a placebo). Randomization minimizes selection bias and confounding variables, while the controlled environment allows researchers to isolate the effect of the experimental factor on the outcome. The ability to compare outcomes between groups under controlled conditions provides the strongest evidence of a direct causal relationship. This aligns with the principles of evidence-based practice, which the CBIC (Certification Board of Infection Control and Epidemiology) emphasizes for infection prevention and control strategies.
Based on this analysis, the randomized-controlled trial (D) is the study design that provides the best evidence of a direct causal relationship. This conclusion is consistent with the CBIC's focus on high-quality evidence to inform infection control practices, as RCTs are prioritized in the hierarchy of evidence for establishing cause-and-effect relationships.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated guidelines, 2023), which emphasizes the use of high-quality evidence, including RCTs, for validating infection control interventions.
CBIC Examination Content Outline, Domain I: Identification of Infectious Disease Processes, which underscores the importance of evidence-based study designs in infection control research.
Passive immunity results from the use of:
Options:
Tetanus antitoxin
Hepatitis B vaccine
Influenza vaccine
Human diploid cell rabies vaccine
Answer:
AExplanation:
The Certification Study Guide (6th edition) defines passive immunity as protection that results from the administration of preformed antibodies, rather than stimulation of the individual’s own immune system. Passive immunity provides immediate but temporary protection, because the recipient does not produce antibodies and therefore does not develop immunologic memory.
Tetanus antitoxin is a classic example of passive immunity. It contains antibodies that neutralize tetanus toxin directly and is used in situations where immediate protection is needed, such as after certain wounds in individuals with unknown or inadequate vaccination history. The study guide emphasizes that passive immunization is particularly important in post-exposure management when waiting for an active immune response would be too slow to prevent disease.
The other options represent active immunization, not passive immunity. Vaccines such as hepatitis B vaccine, influenza vaccine, and human diploid cell rabies vaccine stimulate the recipient’s immune system to produce its own antibodies and immune memory. While rabies immune globulin provides passive immunity, the rabies vaccine itself is an active immunizing agent.
This distinction between active and passive immunity is a frequently tested CIC exam concept, especially in the context of occupational health, post-exposure prophylaxis, and immunization programs. Recognizing that passive immunity involves antibody products (antitoxins or immune globulins) rather than vaccines is essential for accurate infection prevention decision-making.
Which of the following infectious diseases is associated with environmental fungi?
Options:
Listeriosis
Hantavirus
Mucormycosis
Campylobacter
Answer:
CExplanation:
The correct answer is C, "Mucormycosis," as it is the infectious disease associated with environmental fungi. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, mucormycosis is caused by fungi belonging to the order Mucorales, which are commonly found in the environment, including soil, decaying organic matter, and contaminated water. These fungi can become opportunistic pathogens, particularly in immunocompromised individuals, leading to severe infections such as rhinocerebral, pulmonary, or cutaneous mucormycosis (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.1 - Identify infectious disease processes). Environmental exposure, such as inhalation of fungal spores or contact with contaminated materials, is a primary mode of transmission, making it directly linked to environmental fungi.
Option A (Listeriosis) is caused by the bacterium Listeria monocytogenes, typically associated with contaminated food products (e.g., unpasteurized dairy or deli meats) rather than environmental fungi. Option B (Hantavirus) is a viral infection transmitted through contact with rodent excreta, not fungi, and is linked to environmental reservoirs like rodent-infested areas. Option D (Campylobacter) is a bacterial infection caused by Campylobacter species, often associated with undercooked poultry or contaminated water, and is not related to fungi.
The association of mucormycosis with environmental fungi underscores the importance of infection prevention strategies, such as controlling environmental contamination and protecting vulnerable patients, which aligns with CBIC’s focus on identifying and mitigating risks from infectious agents in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). This knowledge is critical for infection preventionists to guide environmental cleaning and patient care protocols.
The infection preventionist notes an increase in Clostridioides difficile infections (CDI) in the ICU. A Root Cause Analysis (RCA) is scheduled. What is the goal of a Root Cause Analysis?
Options:
Proactively identify potential failures.
Identify processes to prevent recurrence.
Determine strengths, weaknesses, opportunities, and threats.
Educate staff in order to avoid individual blame.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) defines a Root Cause Analysis (RCA) as a retrospective, systematic process used to understand why an adverse event or undesired outcome occurred and what system-level changes are needed to prevent it from happening again. In the context of an increase in Clostridioides difficile infections in an ICU, the primary goal of an RCA is to identify underlying process failures and implement corrective actions to prevent recurrence.
RCA focuses on systems and processes rather than individual performance. Through structured methods such as event mapping, cause-and-effect analysis, and contributing factor review, the team examines elements such as antimicrobial use, environmental cleaning practices, hand hygiene compliance, isolation implementation, diagnostic testing practices, and workflow design. The ultimate outcome of an RCA is a set of actionable, sustainable process improvements that reduce the likelihood of similar events in the future.
Option A describes Failure Mode and Effects Analysis (FMEA), which is a proactive risk assessment tool. Option C refers to a SWOT analysis, used for strategic planning rather than event investigation. Option D reflects an important principle of RCA culture (non-punitive), but it is not the primary goal.
For the CIC® exam, it is essential to recognize that the core purpose of RCA is preventing recurrence through system improvement, making option B the correct answer.
==========
During an outbreak investigation of Pseudomonas aeruginosa in a medical intensive care unit (ICU), what is a critical INITIAL step the infection preventionist (IP) should take to better understand an investigation process and this organism?
Options:
Notify public health officials to alert them of the outbreak.
Consult with other IPs in their region to find out what others have seen.
Conduct a literature search that summarizes similar outbreak investigations.
Contact the Centers for Disease Control and Prevention to determine if anyone in their area has experienced similar situations.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that a critical initial step in any outbreak investigation is for the infection preventionist to develop a strong understanding of the organism involved, its epidemiology, reservoirs, modes of transmission, and previously reported outbreak sources. Conducting a literature search of similar outbreak investigations provides this foundational knowledge and helps guide a structured, evidence-based investigation.
Pseudomonas aeruginosa is an opportunistic, water-associated pathogen frequently implicated in healthcare-associated outbreaks, particularly in ICUs. Prior outbreak investigations described in the literature commonly identify sources such as sink drains, faucets, respiratory equipment, humidifiers, contaminated medications, and inadequate reprocessing of medical devices. Reviewing published investigations allows the IP to anticipate likely sources, identify high-yield environmental sampling locations, and avoid unnecessary or unfocused interventions.
Options A and D may become appropriate later, depending on outbreak magnitude and reporting requirements, but they are not the initial step. Option B can be helpful but relies on anecdotal experience rather than systematic evidence. The Study Guide stresses that outbreak investigations should begin with background research and hypothesis generation, followed by targeted data collection and analysis.
For the CIC® exam, this question reinforces that effective outbreak management starts with understanding what is already known, making a literature review the most appropriate initial action.
A healthcare personnel has an acute group A streptococcal throat infection. What is the earliest recommended time that this person may return to work after receiving appropriate antibiotic therapy?
Options:
8 hours
24 hours
48 hours
72 hours
Answer:
BExplanation:
The correct answer is B, "24 hours," as this is the earliest recommended time that a healthcare personnel with an acute group A streptococcal throat infection may return to work after receiving appropriate antibiotic therapy. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC), healthcare workers with group A Streptococcus (GAS) infections, such as streptococcal pharyngitis, should be treated with antibiotics (e.g., penicillin or a suitable alternative) to eradicate the infection and reduce transmission risk. The CDC and Occupational Safety and Health Administration (OSHA) guidelines specify that healthcare personnel can return to work after at least 24 hours of effective antibiotic therapy, provided they are afebrile and symptoms are improving, as this period is sufficient to significantly reduce the bacterial load and contagiousness (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents).
Option A (8 hours) is too short a duration to ensure the infection is adequately controlled and the individual is no longer contagious. Option C (48 hours) and Option D (72 hours) are longer periods that may apply in some cases (e.g., if symptoms persist or in outbreak settings), but they exceed the minimum recommended time based on current evidence. The 24-hour threshold is supported by studies showing that GAS shedding decreases substantially within this timeframe with appropriate antibiotic treatment, minimizing the risk to patients and colleagues (CDC Guidelines for Infection Control in Healthcare Personnel, 2019).
The infection preventionist’s role includes enforcing return-to-work policies to prevent healthcare-associated infections (HAIs), aligning with CBIC’s emphasis on timely and evidence-based interventions to control infectious disease transmission in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). Compliance with this recommendation also supports occupational health protocols to balance staff safety and patient care.
During the past week, three out of four blood cultures from a febrile neonate in an intensive care unit grew coagulase-negative staphylococci. This MOST likely indicates:
Options:
Laboratory error.
Contamination.
Colonization.
Infection.
Answer:
BExplanation:
The scenario involves a febrile neonate in an intensive care unit (ICU) with three out of four blood cultures growing coagulase-negative staphylococci (CoNS) over the past week. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes accurate interpretation of microbiological data in the "Identification of Infectious Disease Processes" domain, aligning with the Centers for Disease Control and Prevention (CDC) guidelines for healthcare-associated infections. Determining whether this represents a true infection, contamination, colonization, or laboratory error requires evaluating the clinical and microbiological context.
Option B, "Contamination," is the most likely indication. Coagulase-negative staphylococci, such as Staphylococcus epidermidis, are common skin flora and frequent contaminants in blood cultures, especially in neonates where skin preparation or sampling technique may be challenging. The CDC’s "Guidelines for the Prevention of Intravascular Catheter-Related Infections" (2017) and the Clinical and Laboratory Standards Institute (CLSI) note that multiple positive cultures (e.g., two or more) are typically required to confirm true bacteremia, particularly with CoNS, unless accompanied by clear clinical signs of infection (e.g., worsening fever, hemodynamic instability) and no other explanation. The inconsistency (three out of four cultures) and the neonate’s ICU setting—where contamination from skin or catheter hubs is common—suggest that the positive cultures likely result from contamination during blood draw rather than true infection. Studies, such as those in the Journal of Clinical Microbiology (e.g., Beekmann et al., 2005), indicate that CoNS in blood cultures is contaminated in 70-80% of cases when not supported by robust clinical correlation.
Option A, "Laboratory error," is possible but less likely as the primary explanation. Laboratory errors (e.g., mislabeling or processing mistakes) could occur, but the repeated growth in three of four cultures suggests a consistent finding rather than a random error, making contamination a more plausible cause. Option C, "Colonization," refers to the presence of microorganisms on or in the body without invasion or immune response. While CoNS can colonize the skin or catheter sites, colonization does not typically result in positive blood cultures unless there is an invasive process, which is not supported by the data here. Option D, "Infection," is the least likely without additional evidence. True CoNS bloodstream infections (e.g., catheter-related) in neonates are serious but require consistent positive cultures, clinical deterioration (e.g., persistent fever, leukocytosis), and often imaging or catheter removal confirmation. The febrile state alone, with inconsistent culture results, does not meet the CDC’s criteria for diagnosing infection (e.g., at least two positive cultures from separate draws).
The CBIC Practice Analysis (2022) and CDC guidelines stress differentiating contamination from infection to avoid unnecessary treatment, which can drive antibiotic resistance. Given the high likelihood of contamination with CoNS in this context, Option B is the most accurate answer.
Based on the Spaulding classification, which of the following pairings is an example of a semi-critical item and its minimal level of disinfection?
Options:
Bedside table; high-level disinfection
Surgical instrument; sterilization
Endocavity probe; high-level disinfection
Bedpan; intermediate-level disinfection
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of applying Spaulding’s classification to determine the appropriate minimum level of processing for medical devices. Under this system, devices are categorized as critical, semi-critical, or noncritical based on the degree of infection risk associated with their use.
Semi-critical items are those that come into contact with mucous membranes or non-intact skin but do not ordinarily penetrate sterile tissue. Examples include endocavity probes, such as transvaginal or transrectal ultrasound probes. Because mucous membranes are more susceptible to infection than intact skin, semi-critical items require at least high-level disinfection after thorough cleaning to eliminate all microorganisms except large numbers of bacterial spores.
Option C correctly pairs an endocavity probe with high-level disinfection, which is the minimum acceptable level of processing for this classification. Option A is incorrect because a bedside table is a noncritical item and requires only low-level disinfection. Option B describes a critical item, which correctly requires sterilization but does not meet the question’s focus on semi-critical devices. Option D is incorrect because bedpans are noncritical items, and intermediate-level disinfection exceeds the minimum requirement.
Understanding Spaulding’s classification and matching devices to the correct level of disinfection is a high-yield topic on the CIC® exam and essential for safe infection prevention practice.
==========
A nutrition support team wants to determine whether patients who receive total parenteral nutrition (TPN) at home are at increased risk of central line–associated bloodstream infection (CLABSI) compared with patients who receive TPN in the hospital. The BEST way to compare these two groups is to calculate the:
Options:
Percentage of patients in each group who became infected.
Infections per 1,000 central line days in each group.
Number of infections in each group this year compared to last year.
Ratio of infected to noninfected central lines in each group.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that accurate comparison of healthcare-associated infection risk between groups requires use of standardized, exposure-based rates. For central line–associated bloodstream infections (CLABSIs), the recommended metric is infections per 1,000 central line days, which accounts for the amount of time patients are actually exposed to the risk factor—in this case, the presence of a central venous catheter.
Patients receiving TPN at home and those receiving TPN in the hospital may differ substantially in duration of catheter use, care practices, and patient acuity. Simply comparing percentages or raw numbers of infections fails to adjust for differences in central line utilization and can result in misleading conclusions. By using central line days as the denominator, infection rates are normalized and allow for valid comparisons between populations and settings.
Option A does not account for differences in exposure time. Option C compares different time periods rather than comparing risk between groups. Option D provides a ratio but lacks standardization and is not consistent with accepted surveillance methodology.
The Study Guide reinforces that device-associated infection surveillance—such as CLABSI monitoring—must use device days to assess true risk and guide prevention strategies. Understanding and applying correct epidemiologic measures is a core competency for infection preventionists and a frequently tested concept on the CIC® exam.
==========
During a COVID outbreak with hospital-associated transmission cases, the infection preventionist (IP) receives a news media call about what is being done to reduce the transmission. The IP's BEST response is to
Options:
answer the questions truthfully.
give vague answers to ensure patient privacy.
refer the reporters to the hospital's media spokesperson.
inform the reporter that the conversation must be recorded to ensure accuracy.
Answer:
CExplanation:
The best response for an infection preventionist (IP) when receiving a news media call during a COVID outbreak with hospital-associated transmission cases is to refer the reporters to the hospital's media spokesperson. This approach aligns with the principles outlined in the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which emphasize the importance of maintaining professionalism, protecting patient privacy, and ensuring accurate communication. The IP's primary role is to focus on infection prevention and control activities rather than serving as a public relations representative. Engaging directly with the media can risk divulging sensitive patient information or operational details that may not be fully contextualized, potentially violating the Health Insurance Portability and Accountability Act (HIPAA) or other privacy regulations.
Option A (answer the questions truthfully) is not ideal because, while truthfulness is important, the IP may not have the authority or full context to provide a comprehensive and accurate public statement, and doing so could inadvertently compromise patient confidentiality or misrepresent hospital policies. Option B (give vague answers to ensure patient privacy) might protect privacy but could lead to miscommunication or lack of trust if the responses appear evasive without a clear referral process. Option D (inform the reporter that the conversation must be recorded to ensure accuracy) is a procedural step but does not address the core issue of who should handle media inquiries.
Referring to the hospital's media spokesperson (Option C) ensures that a trained individual handles the communication, adhering to CBIC's emphasis on collaboration with organizational leadership and adherence to institutional communication protocols (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This also aligns with best practices for managing public health crises, where centralized and coordinated messaging is critical to avoid misinformation.
Which of the following procedures has NOT been documented to contribute to the development of postoperative infections in clean surgical operations?
Options:
Prolonged preoperative hospital stay
Prolonged length of the operations
The use of iodophors for preoperative scrubs
Shaving the site on the day prior to surgery
Answer:
CExplanation:
Postoperative infections in clean surgical operations, defined by the Centers for Disease Control and Prevention (CDC) as uninfected operative wounds with no inflammation and no entry into sterile tracts (e.g., gastrointestinal or respiratory systems), are influenced by various perioperative factors. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes identifying and mitigating risk factors in the "Prevention and Control of Infectious Diseases" domain, aligning with CDC guidelines for surgical site infection (SSI) prevention. The question focuses on identifying a procedure not documented as a contributor to SSIs, requiring an evaluation of evidence-based risk factors.
Option C, "The use of iodophors for preoperative scrubs," has not been documented to contribute to the development of postoperative infections in clean surgical operations. Iodophors, such as povidone-iodine, are antiseptic agents used for preoperative skin preparation and surgical hand scrubs. The CDC’s "Guideline for Prevention of Surgical Site Infections" (1999) and its 2017 update endorse iodophors as an effective method for reducing microbial load on the skin, with no evidence suggesting they increase SSI risk when used appropriately. Studies, including those cited by the CDC, show that iodophors are comparable to chlorhexidine in efficacy for preoperative antisepsis, and their use is a standard, safe practice rather than a risk factor.
Option A, "Prolonged preoperative hospital stay," is a well-documented risk factor. Extended hospital stays prior to surgery increase exposure to healthcare-associated pathogens, raising the likelihood of colonization and subsequent SSI, as noted in CDC and surgical literature (e.g., Mangram et al., 1999). Option B, "Prolonged length of the operations," is also a recognized contributor. Longer surgical durations are associated with increased exposure time, potential breaches in sterile technique, and higher infection rates, supported by CDC data showing a correlation between operative time and SSI risk. Option D, "Shaving the site on the day prior to surgery," has been documented as a risk factor. Preoperative shaving, especially with razors, can cause microabrasions that serve as entry points for bacteria, increasing SSI rates. The CDC recommends avoiding shaving or using clippers immediately before surgery to minimize this risk, with evidence from studies like those in the 1999 guideline showing higher infection rates with preoperative shaving.
The CBIC Practice Analysis (2022) and CDC guidelines focus on evidence-based practices, and the lack of documentation linking iodophor use to increased SSIs—coupled with its role as a preventive measure—makes Option C the correct answer. The other options are supported by extensive research as contributors to SSI development in clean surgeries.
Which of the following is the BEST study design for assessing the benefit of a new treatment?
Options:
Interrupted time series
Correlational study
Parallel group study
Randomized controlled trial
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the randomized controlled trial (RCT) as the gold standard study design for assessing the benefit of a new treatment. RCTs are specifically designed to determine causality by minimizing bias and confounding variables through random assignment of participants to intervention and control groups. This ensures that differences in outcomes can be attributed with the highest level of confidence to the treatment being studied rather than to external factors.
In an RCT, participants are randomly allocated to receive either the new treatment or a comparison intervention (such as standard therapy or placebo). Randomization balances known and unknown risk factors between groups, while controlled conditions allow precise measurement of treatment effects. This design is particularly important when evaluating new therapies, medications, or interventions where efficacy and safety must be clearly demonstrated.
The other study designs listed are less rigorous for assessing treatment benefit. An interrupted time series is useful for evaluating system-level interventions over time but is more susceptible to confounding influences. A correlational study can identify associations but cannot establish cause and effect. A parallel group study without randomization lacks adequate control for bias and confounding.
For CIC® exam preparation, it is essential to recognize that when the objective is to assess the benefit or effectiveness of a new treatment, a randomized controlled trial provides the strongest and most reliable evidence, making it the best answer.
A 17-year-old presents to the Emergency Department with fever, stiff neck, and vomiting. A lumbar puncture is done. The Gram stain shows Gram negative diplocooci. Presumptive identification of the organism is
Options:
Haemophilus influenzae
Neisseria meningitidis
Listeria monocytogenes
Streptococcus pneumoniae
Answer:
BExplanation:
The Gram stain showing Gram-negative diplococci in cerebrospinal fluid (CSF) is characteristic of Neisseria meningitidis, a leading cause of bacterial meningitis in adolescents and young adults.
Step-by-Step Justification:
Gram Stain Interpretation:
Gram-negative diplococci in CSF strongly suggest Neisseria meningitidis.
Classic Symptoms of Meningitis:
Fever, stiff neck, and vomiting are hallmark signs of meningococcal meningitis.
Neisseria meningitidis vs. Other Bacteria:
Haemophilus influenzae (Option A) → Gram-negative coccobacilli.
Listeria monocytogenes (Option C) → Gram-positive rods.
Streptococcus pneumoniae (Option D) → Gram-positive diplococci.
CBIC Infection Control References:
APIC Ready Reference for Microbes, "Neisseria meningitidis and Meningitis".
A director of the Infection Prevention and Control Department has received permission to hire more staff to a total of 3.8 full-time equivalents (FTEs), based on 40 hours/week. The director works 40 hours/week, one infection preventionist (IP) works 32 hours/week, another works 16 hours/week, and secretarial support works 40 hours/week. How many additional FTEs may the director hire?
Options:
0.4
0.6
0.8
1.0
Answer:
BExplanation:
The Certification Study Guide (6th edition) stresses that infection prevention leaders must understand basic workforce and FTE calculations to ensure appropriate staffing and compliance with approved resource allocations. An FTE is defined as 40 hours worked per week, and part-time hours must be converted proportionally.
First, calculate the FTEs already in use:
Director: 40 hours/week ÷ 40 = 1.0 FTE
Infection preventionist: 32 hours/week ÷ 40 = 0.8 FTE
Infection preventionist: 16 hours/week ÷ 40 = 0.4 FTE
Secretarial support: 40 hours/week ÷ 40 = 1.0 FTE
Total current FTEs:
1.0 + 0.8 + 0.4 + 1.0 = 3.2 FTEs
The approved staffing total is 3.8 FTEs. To determine how many additional FTEs may be hired, subtract current FTE usage from the approved total:
3.8 − 3.2 = 0.6 FTE
Therefore, the director may hire 0.6 additional FTE, which could be fulfilled by a part-time infection preventionist or split among staff roles, depending on organizational needs.
CIC exam questions frequently test practical management skills, including staffing calculations, budgeting awareness, and resource allocation. Accurate FTE calculations ensure compliance with administrative approvals and support safe, effective infection prevention program operations.
Which of the following BEST describes the content of an interpretive surveillance report?
Options:
Outlines the organization’s mission for patient quality and safety
Cites the frequency of the evaluation of the monitoring program
Highlights the steps of the facility’s quality improvement activities
Provides findings in a manner designed for the intended audience
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that an interpretive surveillance report goes beyond simply presenting raw data. Its primary purpose is to translate surveillance findings into meaningful, actionable information that can be understood and used by the intended audience, such as frontline staff, clinical leaders, executive leadership, or quality committees.
Interpretive reports contextualize infection data by explaining trends, comparisons, implications, and recommended actions. This may include highlighting increases or decreases in infection rates, identifying areas of concern, interpreting statistical significance, and linking findings to prevention strategies. The format, level of detail, and language are tailored to the audience’s role and decision-making responsibilities. For example, senior leadership may need high-level summaries and risk implications, while unit-level staff benefit from detailed, practice-focused feedback.
Option A describes a mission statement, not a surveillance report. Option B refers to program evaluation logistics rather than interpretation of findings. Option C outlines quality improvement processes but does not describe how surveillance data are communicated.
For the CIC® exam, it is essential to recognize that interpretive surveillance reporting focuses on meaningful communication, not just data display. Providing findings in a manner designed for the intended audience ensures surveillance data drive prevention actions, accountability, and performance improvement—making option D the best answer.
Which of the following options describes a best practice for avoiding occupational health hazards in a healthcare facility?
Options:
The facility is required to conduct annual tuberculosis (TB) testing for healthcare personnel.
The facility is required to conduct varicella post-vaccination testing for healthcare personnel.
The facility should exclude asymptomatic Bordetella pertussis–exposed healthcare personnel from duty.
The facility should provide Neisseria meningitidis pre-exposure vaccination to laboratory personnel.
Answer:
DExplanation:
The Certification Study Guide (6th edition) emphasizes that occupational health hazard prevention is based on risk assessment and targeted protection strategies, particularly for personnel with predictable, high-risk exposures. Providing pre-exposure vaccination against Neisseria meningitidis to laboratory personnel is a recognized best practice because laboratorians who routinely handle N. meningitidis isolates are at increased risk for aerosol or droplet exposure, which can result in rapidly progressive and potentially fatal disease.
The study guide highlights that pre-exposure immunization is preferred over post-exposure management when exposure risk is ongoing and well defined. This strategy aligns with evidence-based occupational health principles and recommendations from public health authorities, making it a proactive and preventive measure rather than a reactive one.
The other options are incorrect because they either reflect outdated practices or inappropriate control measures. Routine annual TB testing is no longer universally required and should be based on facility risk assessment. Post-vaccination varicella serologic testing is not recommended because commercial assays may not reliably detect vaccine-induced immunity. Excluding asymptomatic pertussis-exposed healthcare personnel from duty is not routinely recommended if appropriate prophylaxis is provided.
This question reflects a common CIC exam theme: best practices focus on targeted, evidence-based prevention, especially vaccination strategies for high-risk occupational groups.
An infection preventionist (IP) meets with the operating room staff to discuss an increased number of patients with infections caused by different organisms after joint replacement surgeries. After reviewing case records, the operating room staff members note compliance with operating room standards. Which of the following options BEST explains this discrepancy?
Options:
The operating room data collectors are inexperienced.
The IP is unfamiliar with operating room processes.
Process indicators may not explain outcomes.
The time frames for the data collection are different.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) clearly differentiates between process measures and outcome measures in infection prevention and quality improvement. Process indicators measure whether specific practices or standards are being followed, such as adherence to operating room protocols, environmental controls, or sterile technique. Outcome indicators, on the other hand, reflect the end result, such as the occurrence of surgical site infections (SSIs).
In this scenario, operating room staff demonstrate compliance with established standards, yet an increase in post–joint replacement infections is observed. This discrepancy is best explained by the principle that process compliance alone does not guarantee desired outcomes. Even when processes appear to be correctly followed, infections may still occur due to factors outside the measured processes, such as patient-related risk factors, organism virulence, antimicrobial resistance, or unmeasured system variables.
Options A and B incorrectly focus on personnel competency rather than measurement limitations. Option D may affect data interpretation but does not explain why compliant processes fail to correlate with outcomes. The Study Guide emphasizes that outcome measures are influenced by multiple interacting variables, and therefore a single set of process indicators may not fully explain infection trends.
For the CIC® exam, it is critical to understand that process measures support improvement but do not always predict outcomes, highlighting the need for comprehensive analysis when infection rates rise despite apparent compliance.
Surgical site infection (SSI) data for the previous quarter reveal the following numbers. The surgeon with the highest infection rate is Doctor
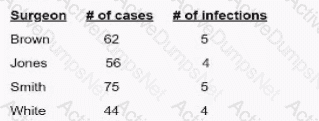
Options:
Brown
Jones.
Smith
White
Answer:
DExplanation:
To determine which surgeon has the highest surgical site infection (SSI) rate, use the following formula:
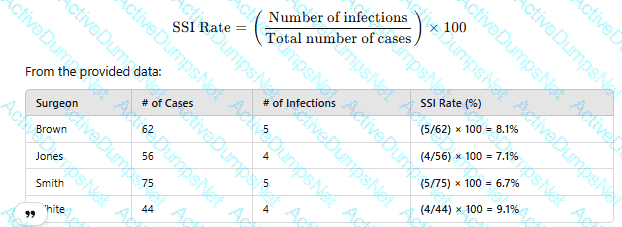
Since Dr. White has the highest SSI rate at 9.1%, the correct answer is D. White.
CBIC Infection Control Reference
SSI rates are calculated using infection count per total procedures and reported as percentage values.
A patient has a draining sinus at the site of a left total hip arthroplasty. A culture from the sinus tract reveals four organisms. Which of the following specimens is optimal for identifying the eliologic agent?
Options:
Blood
Wound drainage
Joint aspirate
Sinus tract tissue
Answer:
CExplanation:
The optimal specimen for identifying the etiologic agent in a prosthetic joint infection (PJI) is a joint aspirate (synovial fluid). This is because:
It provides direct access to the infected site without contamination from external sources.
It allows for accurate microbiologic culture, Gram stain, and leukocyte count analysis.
Why the Other Options Are Incorrect?
A. Blood – Blood cultures may help detect hematogenous spread but are not the best sample for identifying localized prosthetic joint infections.
B. Wound drainage – Wound cultures often contain contaminants from surrounding skin flora and do not accurately reflect joint space infection.
D. Sinus tract tissue – Cultures from sinus tracts often represent colonization rather than the primary infecting organism.
CBIC Infection Control Reference
APIC guidelines confirm that joint aspirate is the most reliable specimen for diagnosing prosthetic joint infections.
The infection preventionist (IP) is invited to a planning meeting for a new oncology unit. The team is excited about the new design and wants lots of natural plants to be incorporated. What action should the IP take?
Options:
Allow the process to continue.
Ask about the air handling unit.
Ask whether artificial plants could be used instead.
Prohibit any discussion on the inclusion of natural plants.
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) clearly emphasizes that oncology units house highly immunocompromised patients, making environmental sources of infection a critical concern during design and planning phases. Natural plants, soil, and standing water are well-recognized reservoirs for environmental fungi and gram-negative bacteria, including Aspergillus, Fusarium, and Pseudomonas species, all of which pose a serious infection risk to oncology patients.
Rather than allowing the process to continue unchecked (Option A) or completely shutting down discussion (Option D), the infection preventionist’s role is to guide the team toward safer alternatives while supporting collaborative planning. Asking whether artificial plants can be used instead (Option C) is the most appropriate action because it maintains the aesthetic goals of the design team while eliminating the infection risks associated with live plants.
Option B, asking about the air handling unit, is important in oncology design but does not directly address the specific and preventable risk posed by natural plants. The Study Guide notes that potted plants, dried flower arrangements, and soil-containing décor should be avoided in areas caring for severely immunocompromised patients.
For the CIC® exam, this question highlights the IP’s responsibility to anticipate environmental infection risks early in facility planning and recommend practical, evidence-based alternatives that protect patient safety without unnecessarily impeding design goals.
An outbreak of carbapenem-resistant Klebsiella pneumoniae is linked to duodenoscopes. What is the infection preventionist’s PRIORITY intervention?
Options:
Perform targeted patient screening for Klebsiella pneumoniae.
Implement immediate enhanced reprocessing procedures and audit compliance.
Discontinue the use of duodenoscopes until further notice.
Conduct whole-genome sequencing of outbreak isolates.
Answer:
BExplanation:
The CDC and FDA have identified duodenoscopes as high-risk devices due to inadequate reprocessing, leading to MDRO transmission.
The first priority is enhancing reprocessing protocols and ensuring strict compliance with manufacturer instructions.
CBIC Infection Control References:
APIC Text, "Endoscope Reprocessing and Infection Risk," Chapter 10.
Essential knowledge, behaviors, and skills that an individual should possess and demonstrate to practice in a specific discipline defines which of the following?
Options:
Certification
Competence
Knowledge
Training
Answer:
BExplanation:
The correct answer is B, "Competence," as it defines the essential knowledge, behaviors, and skills that an individual should possess and demonstrate to practice in a specific discipline. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, competence encompasses the integrated application of knowledge, skills, and behaviors required to perform effectively in a professional role, such as infection prevention and control. Competence goes beyond mere knowledge or training by including the ability to apply these attributes in real-world scenarios, ensuring safe and effective practice (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.3 - Assess competence of healthcare personnel). This holistic definition is critical in healthcare settings, where demonstrated competence—through actions like proper hand hygiene or outbreak management—directly impacts patient safety and infection prevention outcomes.
Option A (certification) refers to a formal recognition or credential (e.g., CIC certification) that validates an individual’s qualifications, but it is an outcome or process rather than the definition of the underlying abilities. Option C (knowledge) represents the theoretical understanding or factual basis of a discipline, which is a component of competence but not the full scope that includes behaviors and skills. Option D (training) involves the education or instruction provided to develop skills and knowledge, serving as a means to achieve competence rather than defining it.
The focus on competence aligns with CBIC’s emphasis on ensuring that healthcare personnel are equipped to meet the demands of infection prevention through a combination of education, practice, and evaluation (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This definition supports the development of professionals who can adapt and perform effectively in dynamic healthcare environments.
Microfiber cloths and mops are preferred over cotton because microfiber:
Options:
Is more cost effective.
Is positively charged to better attract dirt.
Can be laundered and dried with other textiles.
Is versatile for both smooth and rough surfaces.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that microfiber cleaning materials are preferred over traditional cotton cloths and mops because of their electrostatic properties, which enhance cleaning effectiveness. Microfiber is composed of very fine synthetic fibers that become positively charged, allowing them to attract and trap negatively charged dirt, dust, and microorganisms rather than simply pushing them across surfaces.
This electrostatic attraction enables microfiber to remove a significantly higher percentage of bacteria and organic material from surfaces compared to cotton, even when used with less cleaning solution or disinfectant. The split fiber structure also increases surface area, allowing microorganisms and debris to be captured within the fibers rather than redistributed. These properties make microfiber particularly effective for environmental cleaning in healthcare settings, where surface contamination contributes to transmission of healthcare-associated infections.
Option A is incorrect because microfiber products are often more expensive initially, though they may be cost-effective over time. Option C is incorrect because microfiber must be laundered separately under specific conditions to maintain effectiveness. Option D may be true but is not the primary reason for preference.
For the CIC® exam, it is important to recognize that microfiber’s positive charge and superior ability to attract and retain microorganisms are the key reasons it is favored over cotton for environmental cleaning and infection prevention.
A city has a population of 150.000. Thirty new cases of tuberculosis (TB) were diagnosed in the city last year. These now cases brought the total number of active TB cases in the city last year to 115. Which of the following equations represents the incidence rate tor TB per 100.000 in that year?
Options:
(30 ÷ 150.000) x 100.000 = X
(30÷ 150.000) x 100 = X
(115 ÷ 150.000) x 100.000 - X
(115 ÷ 100.000) x 100 = X
Answer:
AExplanation:
The incidence rate is calculated using the formula:

Why the Other Options Are Incorrect?
B. (30 ÷ 150,000) × 100 = X – Incorrect multiplier (should be 100,000 for standard incidence rate).
C. (115 ÷ 150,000) × 100,000 = X – 115 represents total cases (prevalence), not incidence.
D. (115 ÷ 100,000) × 100 = X – Uses the wrong denominator and multiplier.
CBIC Infection Control Reference
APIC defines the incidence rate as the number of new cases per population unit, typically per 100,000 people.
Following an outbreak of Hepatitis A, the water supply is sampled. A high count of which of the following isolates would indicate that the water was a potential source?
Options:
Coliforms
Pseudomonads
Legionella
Acinetobacter
Answer:
AExplanation:
Coliform bacteria are indicators of fecal contamination in water, making them a critical measure of water safety. Hepatitis A is a virus primarily transmitted via the fecal-oral route, often through contaminated food or water.
Step-by-Step Justification:
Fecal Contamination and Hepatitis A:
Hepatitis A virus (HAV) spreads through ingestion of water contaminated with fecal matter. High coliform counts indicate fecal contamination and increase the risk of HAV outbreaks.
Use of Coliforms as Indicators:
Public health agencies use total coliforms and Escherichia coli (E. coli) as primary indicators of water safety because they signal fecal pollution.
Waterborne Transmission of Hepatitis A:
Hepatitis A outbreaks have been traced to contaminated drinking water, ice, and improperly treated wastewater. Coliform detection signals a need for immediate action.
Why Other Options Are Incorrect:
B. Pseudomonads:
Pseudomonads (e.g., Pseudomonas aeruginosa) are environmental bacteria but are not indicators of fecal contamination.
C. Legionella:
Legionella species cause Legionnaires' disease through inhalation of contaminated aerosols, not through fecal-oral transmission.
D. Acinetobacter:
Acinetobacter species are opportunistic pathogens in healthcare settings but are not indicators of waterborne fecal contamination.
CBIC Infection Control References:
APIC Text, "Water Systems and Infection Control Measures".
APIC Text, "Hepatitis A Transmission and Waterborne Outbreaks".
An infection preventionist is developing training exercises for emergency preparedness and disaster response teams. The MOST effective instructional method for retaining information is:
Options:
Providing reading materials to the group.
Watching videos recorded by other hospitals.
Simulating an event to practice how to respond.
Administering a post-test after circulating the emergency response plan.
Answer:
CExplanation:
The Certification Study Guide (6th edition) emphasizes that active, experiential learning methods are the most effective for long-term retention of knowledge and skills, particularly in the context of emergency preparedness and disaster response. Simulation-based training allows participants to practice real-time decision-making, communication, and task execution in a controlled environment that closely mirrors actual emergency conditions.
Simulating an event—such as a mass casualty incident, infectious disease outbreak, or evacuation—engages learners cognitively, physically, and emotionally. The study guide notes that this type of hands-on training improves recall, reinforces correct behaviors, exposes system gaps, and builds team confidence. Simulation also supports interdisciplinary coordination and allows immediate feedback and debriefing, which further enhances learning retention.
The other instructional methods are less effective for retention. Reading materials and watching videos are passive learning approaches that may increase awareness but do not ensure competency during high-stress situations. Administering a post-test measures short-term knowledge acquisition but does not demonstrate the ability to apply that knowledge during an actual emergency.
CIC exam questions frequently highlight adult learning principles, stressing that people learn best by doing—especially when preparing for rare but high-risk events. Simulation-based exercises are therefore considered the gold standard for emergency preparedness training and are strongly recommended for disaster response teams.
Which of the following reasons BEST describes the importance of documenting cleaning, disinfection, and sterilization processes?
Options:
Reduce the cost of hospital operations.
Ensure compliance with Spaulding classification scheme.
Ensure that all processes are conducted on a regular basis.
Comply with policies, regulations, and accreditation standards.
Answer:
DExplanation:
The Certification Study Guide (6th edition) emphasizes that documentation of cleaning, disinfection, and sterilization processes is a fundamental requirement for regulatory compliance and patient safety assurance. Accurate and complete documentation demonstrates that reprocessing activities are performed according to established policies, manufacturer instructions for use (IFUs), and evidence-based standards. This documentation is essential for meeting expectations set by regulatory agencies, accrediting bodies, and internal quality assurance programs.
Documentation provides verifiable proof that critical steps—such as cleaning, monitoring of sterilization parameters, load release, and equipment maintenance—have been performed correctly. In the event of a healthcare-associated infection investigation, recall, or survey, records serve as objective evidence that proper reprocessing practices were followed. The study guide highlights that “if it is not documented, it is considered not done”, a principle commonly tested on the CIC exam.
The other options reflect secondary or indirect benefits but do not represent the primary reason for documentation. Cost reduction is not the intent of reprocessing records. While Spaulding classification informs how items should be reprocessed, documentation alone does not ensure compliance with that framework. Ensuring processes occur regularly is an operational issue rather than a documentation purpose.
CIC exam questions frequently reinforce that documentation supports accountability, traceability, regulatory compliance, and accreditation readiness, making compliance with policies, regulations, and standards the best answer.
Which of the following factors increases a patient’s risk of developing ventilator-associated pneumonia (VAP)?
Options:
Hypoxia
Nasogastric tube
Acute lung disease
In-line suction
Answer:
BExplanation:
Ventilator-associated pneumonia (VAP) is a type of healthcare-associated pneumonia that occurs in patients receiving mechanical ventilation for more than 48 hours. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes identifying risk factors for VAP in the "Prevention and Control of Infectious Diseases" domain, aligning with the Centers for Disease Control and Prevention (CDC) guidelines for preventing ventilator-associated events. The question requires identifying which factor among the options increases a patient’s risk of developing VAP, based on evidence from clinical and epidemiological data.
Option B, "Nasogastric tube," is the correct answer. The presence of a nasogastric tube is a well-documented risk factor for VAP. This tube can facilitate the aspiration of oropharyngeal secretions or gastric contents into the lower respiratory tract, bypassing natural defense mechanisms like the epiglottis. The CDC’s "Guidelines for Preventing Healthcare-Associated Pneumonia" (2004) and studies in the American Journal of Respiratory and Critical Care Medicine (e.g., Kollef et al., 2005) highlight that nasogastric tubes increase VAP risk by promoting microaspiration, especially if improperly managed or if the patient has impaired gag reflexes. This mechanical disruption of the airway’s protective barriers is a direct contributor to infection.
Option A, "Hypoxia," refers to low oxygen levels in the blood, which can be a consequence of lung conditions or VAP but is not a primary risk factor for developing it. Hypoxia may indicate underlying respiratory compromise, but it does not directly increase the likelihood of VAP unless associated with other factors (e.g., prolonged ventilation). Option C, "Acute lung disease," is a broad term that could include conditions like acute respiratory distress syndrome (ARDS), which may predispose patients to VAP due to prolonged ventilation needs. However, acute lung disease itself is not a specific risk factor; rather, it is the need for mechanical ventilation that elevates risk, making this less direct than the nasogastric tube effect. Option D, "In-line suction," involves a closed-system method for clearing respiratory secretions, which is designed to reduce VAP risk by minimizing contamination during suctioning. The CDC and evidence-based guidelines (e.g., American Thoracic Society, 2016) recommend in-line suction to prevent infection, suggesting it decreases rather than increases VAP risk.
The CBIC Practice Analysis (2022) and CDC guidelines prioritize identifying modifiable risk factors like nasogastric tubes for targeted prevention strategies (e.g., elevating the head of the bed to reduce aspiration). Option B stands out as the factor most consistently linked to increased VAP risk based on clinical evidence.
A team was created to determine what has contributed to the recent increase in catheter associated urinary tract infections (CAUTIs). What quality tool should the team use?
Options:
Gap analysis
Fishbone diagram
Plan, do, study, act (PDSA)
Failure mode and effect analysis (FMEA)
Answer:
BExplanation:
The correct answer is B, "Fishbone diagram," as this is the most appropriate quality tool for the team to use when determining what has contributed to the recent increase in catheter-associated urinary tract infections (CAUTIs). According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, the fishbone diagram, also known as an Ishikawa or cause-and-effect diagram, is a structured tool used to identify and categorize potential causes of a problem. In this case, the team needs to explore the root causes of the CAUTI increase, which could include factors such as improper catheter insertion techniques, inadequate maintenance, staff training gaps, or environmental issues (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.2 - Analyze surveillance data). The fishbone diagram organizes these causes into categories (e.g., people, process, equipment, environment), facilitating a comprehensive analysis and guiding further investigation or intervention.
Option A (gap analysis) is useful for comparing current performance against a desired standard or benchmark, but it is more suited for identifying deficiencies in existing processes rather than uncovering the specific causes of a recent increase. Option C (plan, do, study, act [PDSA]) is a cyclical quality improvement methodology for testing and implementing changes, which would be relevant after identifying causes and designing interventions, not as the initial tool for root cause analysis. Option D (failure mode and effect analysis [FMEA]) is a proactive risk assessment tool used to predict and mitigate potential failures in a process before they occur, making it less applicable to analyzing an existing increase in CAUTIs.
The use of a fishbone diagram aligns with CBIC’s emphasis on using data-driven tools to investigate and address healthcare-associated infections (HAIs) like CAUTIs, supporting the team’s goal of pinpointing contributory factors (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.3 - Identify risk factors for healthcare-associated infections). This tool’s visual and collaborative nature also fosters team engagement, which is essential for effective problem-solving in infection prevention.
An infection preventionist has been asked to consult on disinfectant products for use in a long term care home. What should their primary concern be?
Options:
Patient care items are cleaned whenever visibly soiled.
An appropriate disinfectant should be available whenever items are used on patients known to be colonized with multi drug resistant organisms.
Disinfectant products should be compatible with the patient care devices used by the facility.
Disinfectant products should have a mild odor to reduce allergy concerns.
Answer:
CExplanation:
The most critical factor in choosing disinfectants in long-term care is compatibility with medical devices to prevent damage and ensure safety. Improper selection can compromise disinfection efficacy and equipment longevity.
The APIC/JCR Workbook highlights:
“Organizations should evaluate compatibility of disinfectant products with the materials used in patient care equipment. Incompatibility can lead to equipment degradation or malfunction”.
This ensures compliance with manufacturer instructions and preserves warranty and functionality.
Which humoral antibody indicates previous infection and assists in protecting tissue?
Options:
IgA
IgD
IgG
IgM
Answer:
CExplanation:
Humoral antibodies, or immunoglobulins, play distinct roles in the immune system, and their presence or levels can provide insights into infection history and ongoing immune protection. The Certification Board of Infection Control and Epidemiology (CBIC) recognizes the importance of understanding immunological responses in the "Identification of Infectious Disease Processes" domain, which is critical for infection preventionists to interpret diagnostic data and guide patient care. The question focuses on identifying the antibody that indicates a previous infection and assists in protecting tissue, requiring an evaluation of the functions and kinetics of the five major immunoglobulin classes (IgA, IgD, IgG, IgM, IgE).
Option C, IgG, is the correct answer. IgG is the most abundant antibody in serum, accounting for approximately 75-80% of total immunoglobulins, and is the primary antibody involved in long-term immunity. It appears in significant levels after an initial infection, typically rising during the convalescent phase (weeks to months after exposure) and persisting for years, serving as a marker of previous infection. IgG provides protection by neutralizing pathogens, opsonizing them for phagocytosis, and activating the complement system, which helps protect tissues from further damage. The Centers for Disease Control and Prevention (CDC) and clinical immunology references, such as the "Manual of Clinical Microbiology" (ASM Press), note that IgG seroconversion or elevated IgG titers are commonly used to diagnose past infections (e.g., measles, hepatitis) and indicate lasting immunity. Its ability to cross the placenta also aids in protecting fetal tissues, reinforcing its protective role.
Option A, IgA, is primarily found in mucosal secretions (e.g., saliva, tears, breast milk) and plays a key role in mucosal immunity, preventing pathogen adhesion to epithelial surfaces. While IgA can indicate previous mucosal infections and offers localized tissue protection, it is not the primary systemic marker of past infection or long-term tissue protection, making it less fitting. Option B, IgD, is present in low concentrations and is mainly involved in B-cell activation and maturation, with no significant role in indicating previous infection or protecting tissues. Option D, IgM, is the first antibody produced during an acute infection, appearing early in the immune response (within days) and indicating current or recent infection. However, its levels decline rapidly, and it does not persist to mark previous infection or provide long-term tissue protection, unlike IgG.
The CBIC Practice Analysis (2022) and CDC guidelines on serological testing emphasize IgG’s role in assessing past immunity, supported by immunological literature (e.g., Janeway’s Immunobiology, 9th Edition). Thus, IgG is the humoral antibody that best indicates previous infection and assists in protecting tissue, making Option C the correct choice.
Healthcare workers are MOST likely to benefit from infection prevention education if the Infection Preventionist (IP)
Options:
brings in speakers who are recognized experts.
plans the educational program well ahead of time.
audits practices and identifies deficiencies.
involves the staff in determining the content.
Answer:
DExplanation:
The correct answer is D, "involves the staff in determining the content," as this approach is most likely to benefit healthcare workers from infection prevention education. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective education programs are tailored to the specific needs and contexts of the learners. Involving staff in determining the content ensures that the educational material addresses their real-world challenges, knowledge gaps, and interests, thereby increasing engagement, relevance, and application of the learned principles (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). This participatory approach fosters ownership and accountability among healthcare workers, enhancing the likelihood that they will adopt and sustain infection prevention practices.
Option A (brings in speakers who are recognized experts) can enhance credibility and provide high-quality information, but it does not guarantee that the content will meet the specific needs of the staff unless their input is considered. Option B (plans the educational program well ahead of time) is important for logistical success and preparedness, but without staff involvement, the program may lack relevance or fail to address immediate concerns. Option C (audits practices and identifies deficiencies) is a valuable step in identifying areas for improvement, but it is a diagnostic process rather than a direct educational strategy; education based solely on audits might not engage staff effectively if their input is not sought.
The focus on involving staff aligns with CBIC’s emphasis on adult learning principles, which highlight the importance of learner-centered education. By involving staff, the IP adheres to best practices for adult education, ensuring that the program is practical and tailored, ultimately leading to better outcomes in infection prevention (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach also supports a collaborative culture, which is critical for sustaining infection control efforts in healthcare settings.
An HBsAb-negative employee has a percutaneous exposure to blood from a Hepatitis B surface antigen (HBsAg) positive patient. Which of the following regimens is recommended for this employee?
Options:
Immune serum globulin and hepatitis B vaccine
Hepatitis B immune globulin (HBIG) alone
Hepatitis B vaccine alone
Hepatitis B immune globulin (HBIG) and hepatitis B vaccine
Answer:
DExplanation:
The correct answer is D, "Hepatitis B immune globulin (HBIG) and hepatitis B vaccine," as this is the recommended regimen for an HBsAb-negative employee with a percutaneous exposure to blood from an HBsAg-positive patient. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), post-exposure prophylaxis (PEP) for hepatitis B virus (HBV) exposure depends on the employee’s vaccination status and the source’s HBsAg status. For an unvaccinated or known HBsAb-negative individual (indicating no immunity) exposed to HBsAg-positive blood, the standard PEP includes both HBIG and the hepatitis B vaccine. HBIG provides immediate passive immunity by delivering pre-formed antibodies, while the vaccine initiates active immunity to prevent future infections (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). The HBIG should be administered within 24 hours of exposure (preferably within 7 days), and the first dose of the vaccine should be given concurrently, followed by the complete vaccine series.
Option A (immune serum globulin and hepatitis B vaccine) is incorrect because immune serum globulin (ISG) is a general immunoglobulin preparation and not specific for HBV; HBIG, which contains high titers of anti-HBs, is the appropriate specific immunoglobulin for HBV exposure. Option B (hepatitis B immune globulin [HBIG] alone) is insufficient, as it provides only temporary passive immunity without initiating long-term active immunity through vaccination, which is critical for an unvaccinated individual. Option C (hepatitis B vaccine alone) is inadequate for immediate post-exposure protection, as it takes weeks to develop immunity, leaving the employee vulnerable in the interim.
The recommendation for HBIG and hepatitis B vaccine aligns with CBIC’s emphasis on evidence-based post-exposure management to prevent HBV transmission in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This dual approach is supported by CDC guidelines, which prioritize rapid intervention to reduce the risk of seroconversion following percutaneous exposure (CDC Updated U.S. Public Health Service Guidelines for the Management of Occupational Exposures to HBV, HCV, and HIV, 2013).
A positive biological indicator is reported to the Infection Preventionist (IP) after a sterilizer was used. Which of the following should be done FIRST?
Options:
Check the Central Services employees' technique
Inform the risk manager of the positive indicator
Notify potentially affected patients of exposure to nonsterile equipment
Re-challenge the sterilizer with a second indicator
Answer:
DExplanation:
When a positive biological indicator (BI) is detected, the immediate response is to retest the sterilizer using another BI to confirm results. This helps distinguish between a true sterilization failure and a defective BI.
The CBIC Study Guide advises:
“If there is no indication of abnormalities, then the sterilizer should be tested again in three consecutive cycles using paired biological indicators from different manufacturers.”
Immediate recall is reserved for implant loads or confirmed sterilization failure.
Incorrect responses:
A. Check employee technique may be appropriate later but not as a first step.
B. Informing risk manager or C. Notifying patients occurs only after confirmation of failure.
It is determined that the Infection Prevention and Control Program has inadequate resources to accomplish the required tasks. What is the FIRST step?
Options:
Review studies and recommendations on resource allowances for staffing decisions
Contact hospitals in the region to determine their staffing guidelines
Schedule a meeting with supervisor to discuss current job duties
Update the Infection Prevention and Control Plan
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that when an Infection Prevention and Control (IPC) Program identifies inadequate resources, the first and most critical step is internal assessment and communication. Scheduling a meeting with the supervisor to discuss current job duties allows the infection preventionist to clearly define workload demands, regulatory requirements, and gaps between assigned responsibilities and available resources.
This initial discussion establishes a shared understanding of scope of practice, priority tasks, and compliance obligations, such as surveillance, reporting, education, emergency preparedness, and performance improvement. The Study Guide highlights that resource justification must begin with a clear inventory of required functions versus available staffing, time, and tools. Without this foundational step, subsequent actions—such as benchmarking, literature review, or plan updates—lack context and organizational alignment.
Option A is an important later step, used to support justification once internal expectations and gaps are defined. Option B may provide benchmarking data but should not precede internal role clarification. Option D is premature, as program plans should be updated only after leadership agreement on scope, priorities, and resources.
For CIC® exam preparation, it is essential to recognize that effective advocacy for IPC resources begins with direct supervisor engagement, role clarification, and documentation of unmet needs. This structured approach aligns with leadership principles and ensures that requests for additional resources are credible, data-driven, and organizationally relevant.
The BEST choice for surgical instrument cleaning and material compatibility is a detergent solution with:
Options:
An acidic pH
A neutral pH
Sodium hypochlorite
Quaternary ammonium compounds
Answer:
BExplanation:
The Certification Study Guide (6th edition) emphasizes that the primary goal of surgical instrument cleaning is to remove organic and inorganic soil while preserving the integrity and functionality of the instrument. For this reason, detergents with a neutral pH are considered the best choice for routine surgical instrument cleaning and material compatibility.
Neutral pH detergents are effective at removing blood, tissue, and other organic matter without causing corrosion, pitting, or degradation of metals, plastics, seals, and coatings commonly used in surgical instruments. The study guide notes that repeated exposure to harsh chemical environments can damage instruments, compromise device performance, and shorten instrument lifespan—ultimately affecting patient safety and increasing replacement costs.
Acidic detergents may be used selectively for removal of mineral deposits or water scale but are not appropriate for routine cleaning due to their corrosive potential. Sodium hypochlorite (bleach) is strongly discouraged for surgical instruments because it is highly corrosive and can rapidly damage stainless steel. Quaternary ammonium compounds are low-level disinfectants and are not suitable for cleaning critical or semi-critical medical devices prior to disinfection or sterilization.
This question reflects a high-yield CIC exam principle: effective cleaning must balance soil removal with material compatibility. Neutral pH detergents best meet both requirements and are widely recommended by manufacturers and reprocessing standards for surgical instrumentation.
An infection preventionist is providing education to a group of medical device reprocessing staff on critical steps in cleaning instruments. Which of the following actions is recommended while using washer-disinfector?
Options:
Stack instruments inside the machine
Use circulating water with a pH of 3
Disassemble instruments as much as possible
Close hinged instruments prior to placing in the machine
Answer:
CExplanation:
Best practices for using a washer-disinfector include disassembling instruments and opening hinged instruments to ensure proper cleaning and decontamination.
The APIC Text explains:
“Open hinged instruments and disassemble all instruments… Confirm that spray will be able to reach all loaded items without impedance.”
This ensures water and detergents reach all surfaces. Avoid stacking instruments and ensure proper placement to allow full cleaning.
Two patients in a medical intensive care unit (ICU) and one patient in a surgical ICU have Aspergillus fumigatus cultured from sputum. An exterior construction project was started two weeks ago with demolition of an old office building one week ago. All of the following questions are important for the infection preventionist to ask EXCEPT:
Options:
“Have ventilation filters been changed according to the preventive maintenance schedule during construction?”
“Where are the air-intake units relative to these two ICUs?”
“Are there any healthcare personnel that have cared for both of these patients during their hospitalization?”
“Has Aspergillus fumigatus been cultured from these patients prior to construction?”
Answer:
CExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that Aspergillus infections associated with healthcare settings are most commonly environmentally acquired, particularly during construction, renovation, or demolition activities. Aspergillus fumigatus is an airborne mold, and transmission occurs through inhalation of spores, not via person-to-person contact.
In this scenario, the infection preventionist should focus on air handling systems and environmental controls, which makes options A, B, and D critical questions. Ensuring that ventilation filters are appropriately maintained (Option A) and evaluating the proximity of air-intake units to construction activities (Option B) are essential elements of an Infection Control Risk Assessment (ICRA). Asking whether Aspergillus was present before construction (Option D) helps determine whether this represents a construction-associated cluster rather than baseline colonization.
Option C is the least relevant because healthcare personnel do not transmit Aspergillus between patients. Unlike organisms spread via contact or droplets, Aspergillus spores are ubiquitous in dust and air and are introduced through environmental disruption. Therefore, evaluating shared staff assignments does not contribute meaningfully to identifying the source of exposure.
For CIC® exam preparation, it is critical to remember that construction-associated aspergillosis investigations focus on air quality, ventilation, and environmental controls—not staff transmission pathways.
At a facility with 10.000 employees. 5,000 are at risk for bloodbome pathogen exposure. Over the past five years, 100 of the 250 needlestick injuries involved exposure to bloodborne pathogens, and 2% of exposed employees seroconverted. How many employees became infected?
Options:
1
2
5
10
Answer:
BExplanation:
To determine the number of employees who seroconverted (became infected) after a needlestick exposure, we use the given data:
Total Needlestick Injuries: 250
Needlestick Injuries Involving Bloodborne Pathogens: 100
Seroconversion Rate: 2%
Calculation:
Why Other Options Are Incorrect:
A. 1: Incorrect calculation; 2% of 100 is 2, not 1.
C. 5: Overestimates the actual number of infections.
D. 10: Exceeds the calculated value based on given data.
CBIC Infection Control References:
APIC Text, "Occupational Exposure and Seroconversion Risks".
APIC Text, "Bloodborne Pathogens and Needlestick Injury Prevention"
A ventilator-associated pneumonia rate in the ICU has increased from 8.1 infections/1,000 ventilator days to 15.4 infections/1,000 ventilator days over the past two months. To determine the root cause for this increase, the MOST appropriate tool for a performance improvement team is a:
Options:
Fishbone diagram
Pareto chart
Flow diagram
Control chart
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the fishbone diagram, also known as a cause-and-effect diagram or Ishikawa diagram, as the most appropriate tool for conducting root cause analysis when investigating an increase in adverse outcomes such as ventilator-associated pneumonia (VAP). This tool is specifically designed to systematically explore multiple contributing factors that may be driving a problem.
A fishbone diagram helps a multidisciplinary performance improvement team organize potential causes into logical categories, commonly including people, processes, equipment, environment, materials, and policies. In the case of rising VAP rates, the team might examine factors such as ventilator care practices, oral hygiene compliance, head-of-bed elevation, sedation practices, staffing levels, equipment maintenance, and adherence to prevention bundles. By visually mapping these contributors, the team can identify underlying system issues rather than focusing on isolated events or individual performance.
The other tools listed are less appropriate for root cause determination. A Pareto chart is useful for prioritizing the most frequent contributors after causes are identified, but it does not identify causes itself. A flow diagram maps process steps but does not analyze why failures occur. A control chart monitors variation over time but does not explain causation.
For CIC® exam preparation, it is essential to recognize that fishbone diagrams are the primary tool for identifying root causes in performance improvement investigations involving increased infection rates.
A nurse exposed to pertussis develops a mild cough 14 days later. What is the recommended action?
Options:
Continue working with a surgical mask.
Exclude from patient care until five days after starting antibiotics.
Initiate post-exposure prophylaxis only if symptoms worsen.
Conduct serologic testing before deciding on work restrictions.
Answer:
BExplanation:
The CDC recommends exclusion of healthcare workers with pertussis until completing at least five days of antibiotic therapy.
CBIC Infection Control References:
APIC-JCR Workbook, "Occupational Health Considerations," Chapter 10
Which of the following is an example of an outcome measure?
Options:
Hand hygiene compliance rate
Adherence to Environmental Cleaning
Rate of multi-drug resistant organisms acquisition
Timing of preoperative antibiotic administration
Answer:
CExplanation:
The correct answer is C, "Rate of multi-drug resistant organisms acquisition," as it represents an example of an outcome measure. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, outcome measures are indicators that reflect the impact or result of infection prevention and control interventions on patient health outcomes or the incidence of healthcare-associated infections (HAIs). The rate of multi-drug resistant organisms (MDRO) acquisition directly measures the incidence of new infections caused by resistant pathogens, which is a key outcome affected by the effectiveness of infection control practices (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.4 - Evaluate the effectiveness of infection prevention and control interventions).
Option A (hand hygiene compliance rate) is an example of a process measure, which tracks adherence to specific protocols or practices intended to prevent infections, rather than the resulting health outcome. Option B (adherence to environmental cleaning) is also a process measure, focusing on the implementation of cleaning protocols rather than the end result, such as reduced infection rates. Option D (timing of preoperative antibiotic administration) is another process measure, assessing the timeliness of an intervention to prevent surgical site infections, but it does not directly indicate the outcome (e.g., infection rate) of that intervention.
Outcome measures, such as the rate of MDRO acquisition, are critical for evaluating the success of infection prevention programs and are often used to guide quality improvement initiatives. This aligns with CBIC’s emphasis on using surveillance data to assess the effectiveness of interventions and inform decision-making (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies). The focus on MDRO acquisition specifically highlights a significant healthcare challenge, making it a prioritized outcome measure in infection control.
Which of the following is the BEST aid in the identification of patients affected by a recall due to failures in endoscope reprocessing?
Options:
Maintaining a log of endoscope use by date of procedure
Maintaining a log of patient identifiers linked with endoscope used
Searching electronic records for endoscope serial number recorded in patient records
Searching electronic records using diagnostic coding to identify all patients that had endoscopy procedures
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of traceability in endoscope reprocessing programs to ensure rapid and accurate patient notification when reprocessing failures or recalls occur. The most effective method for identifying affected patients is maintaining a log that directly links each endoscope to specific patient identifiers for every procedure.
This type of tracking system allows infection preventionists to quickly determine exactly which patients were exposed to a particular endoscope during the time period of concern. When reprocessing failures are identified—such as incomplete cleaning, high-level disinfection errors, or equipment malfunction—precise linkage between the endoscope and the patient is essential to limit the scope of exposure investigations, reduce unnecessary notifications, and ensure timely follow-up care.
Option A is insufficient because a date-only log does not identify individual patients. Option C may be useful if serial numbers are consistently documented in the medical record, but this practice is not reliably implemented in many facilities and is therefore less dependable. Option D is overly broad and would identify all patients who underwent endoscopy, rather than those exposed to a specific device, leading to unnecessary alarm and inefficient investigations.
For CIC® exam purposes, understanding that patient-to-device linkage logs are the cornerstone of effective exposure investigation and recall management in endoscope reprocessing is critical and aligns with best-practice infection prevention standards.
The infection preventionist observes a nurse obtaining a wound culture and notes which of the following steps is correct?
Options:
The specimen is refrigerated to maintain integrity.
The nurse uses aseptic technique to collect the specimen.
The specimen container is labeled with the patient’s initials.
The specimen is obtained after the antibiotics have been started.
Answer:
BExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that aseptic technique is essential when obtaining clinical specimens, including wound cultures, to ensure accurate results and prevent contamination. Using aseptic technique minimizes the introduction of skin flora or environmental microorganisms that could lead to false-positive cultures and inappropriate clinical management.
Correct wound culture collection includes cleansing the wound as indicated, using sterile equipment, and avoiding contact with surrounding skin or nonsterile surfaces. This approach ensures that organisms identified in the culture are representative of true pathogens rather than contaminants. Proper specimen collection is a foundational infection prevention practice and directly affects diagnostic accuracy, antimicrobial stewardship, and patient outcomes.
Option A is incorrect because wound specimens are typically transported promptly at room temperature; refrigeration is not routinely recommended and may compromise certain organisms. Option C is incorrect because specimen containers must be labeled with at least two patient identifiers (such as full name and medical record number), not initials alone, to meet patient safety standards. Option D is incorrect because specimens should be obtained before initiation of antibiotic therapy whenever possible, as antibiotics can suppress bacterial growth and lead to false-negative results.
For CIC® exam preparation, it is critical to recognize that aseptic technique during specimen collection is the key correct practice, ensuring reliable laboratory results and supporting effective infection prevention and control efforts.
==========
An infection preventionist is writing a policy about prevention of intravascular device infection. Which of the following is important for healthcare personnel to know as part of central line insertion and maintenance procedures?
Options:
Change the central line every seven days.
Use maximum sterile barrier precautions for the line insertion.
The femoral site is the preferred site of insertion in an adult patient.
Use 70% isopropyl alcohol for skin preparation before line insertion.
Answer:
BExplanation:
The Certification Study Guide (6th edition) identifies the use of maximum sterile barrier (MSB) precautions during central line insertion as a cornerstone practice for preventing intravascular device–associated infections, including central line–associated bloodstream infections (CLABSIs). MSB precautions include wearing a cap, mask, sterile gown, and sterile gloves, and using a large sterile drape to fully cover the patient during line insertion. These measures significantly reduce the risk of introducing skin flora and environmental microorganisms into the bloodstream at the time of catheter placement.
The study guide emphasizes that the highest risk for contamination occurs during insertion, making strict aseptic technique essential. MSB precautions are a required element of evidence-based central line insertion bundles and are consistently associated with reduced CLABSI rates when reliably implemented.
The other options reflect outdated or incorrect practices. Routine scheduled replacement of central lines every seven days is not recommended and does not reduce infection risk. The femoral vein is not the preferred insertion site in adults due to higher infection risk compared to subclavian or internal jugular sites. While alcohol is used during hub disinfection, chlorhexidine-based antisepsis (preferably chlorhexidine with alcohol) is recommended for skin preparation—not alcohol alone.
This question highlights a core CIC exam concept: standardized insertion practices using maximum sterile barriers are among the most effective strategies for preventing intravascular device infections.
Which of the following processes would the catheter-associated urinary tract infection (CAUTI) improvement team choose as a process indicator to reduce CAUTI?
Options:
Documentation of indication for catheter placement
Rate of catheter-associated urinary tract infections
Reduction of catheter insertions per month
Rate of asymptomatic bacteriuria
Answer:
AExplanation:
A process indicator measures whether staff are reliably performing evidence-based practices that prevent infection (i.e., how well we do what we intend to do). For CAUTI prevention, a core, guideline-supported strategy is to use indwelling urinary catheters only for appropriate indications and remove them as soon as they are no longer needed. Because inappropriate placement is a major driver of unnecessary catheter days (and therefore CAUTI risk), tracking whether the clinical indication is documented at insertion is a practical, auditable process measure that directly reflects adherence to appropriate-use policies. The CDC CAUTI prevention toolkit lists “compliance with documentation of catheter …” as an example of a process measure, aligning with performance measurement approaches recommended for CAUTI prevention.
In contrast, the CAUTI rate (option B) is an outcome measure, not a process measure. “Reduction of catheter insertions per month” (option C) reflects volume/usage trends rather than direct compliance with a specific practice, and “rate of asymptomatic bacteriuria” (option D) is not a recommended target metric for CAUTI prevention and routine screening for ASB is discouraged in CAUTI guidance.
An infection preventionist (IP) is tasked with identifying if the Intensive Care Unit’s (ICU) central line–associated bloodstream infection (CLABSI) prevention practices are consistent with current best practices. Which of the following quality improvement tools should the IP construct?
Options:
Gap analysis
Root cause analysis
Failure mode and effect analysis (FMEA)
Strengths, weaknesses, opportunities, and threats (SWOT) analysis
Answer:
AExplanation:
The Certification Study Guide (6th edition) clearly distinguishes among quality improvement tools based on their purpose and timing. When the goal is to determine whether current practices align with evidence-based standards or best practices, the most appropriate tool is a gap analysis. A gap analysis systematically compares current state practices—such as ICU CLABSI prevention policies, procedures, and compliance data—with the desired state, which is defined by nationally recognized guidelines and best practices.
The study guide emphasizes that gap analysis is particularly useful for program evaluation, policy review, and baseline assessment before implementing improvements. In this scenario, the IP is not responding to an adverse event, nor is the IP proactively predicting failures, but rather assessing alignment with best practices, which is the core function of a gap analysis.
The other tools serve different purposes. Root cause analysis (RCA) is used after an adverse event (such as a CLABSI) to identify contributing factors. Failure mode and effect analysis (FMEA) is a prospective risk assessment tool used to anticipate where processes might fail. SWOT analysis is a strategic planning tool and is not sufficiently specific for evaluating compliance with infection prevention standards.
Because CIC exam questions frequently test the ability to select the right tool for the right situation, recognizing gap analysis as the appropriate choice in this context is essential.
Which of the following statements is true in considering work reassignment for pregnant employees?
Options:
Pregnant employees rarely require work reassignments
Pregnant employees who are positive for hepatitis B surface antibody may not care for hepatitis B patients
Pregnant employees should not be assigned to patients with known infections
Pregnant employees who are not immune to varicella should be excluded from pediatrics
Answer:
DExplanation:
Pregnant healthcare workers who are not immune to varicella (chickenpox) are at increased risk for severe complications if infected. These employees should be excluded from areas like pediatrics where exposure risk is elevated.
The APIC Text specifies:
“Healthcare personnel who are not immune to varicella should avoid exposure to patients with active disease. In high-risk areas such as pediatrics, nonimmune pregnant employees should be reassigned”.
The CIC Study Guide also supports work exclusion or reassignment of nonimmune pregnant staff who have had exposure to varicella or are at risk.
Explanation of incorrect options:
A. Pregnant employees rarely require reassignment – False; reassignment is required in specific high-risk scenarios.
B. Hepatitis B surface antibody positivity means the employee is immune and can care for HBV patients.
C. Broad exclusion from all infected patients is unnecessary and impractical.
The BEST roommate selection for a patient with active shingles would be a patient who has had
Options:
varicella vaccine.
treatment with acyclovir
a history of herpes simplex.
varicclla zoster immunoglobulin
Answer:
AExplanation:
A patient with active shingles (herpes zoster) is contagious to individuals who have never had varicella (chickenpox) or the varicella vaccine. The best roommate selection is someone who has received the varicella vaccine, as they are considered immune and not at risk for contracting the virus.
Why the Other Options Are Incorrect?
B. Treatment with acyclovir – Acyclovir treats herpes zoster but does not prevent transmission to others.
C. A history of herpes simplex – Prior herpes simplex virus (HSV) infection does not confer immunity to varicella-zoster virus (VZV).
D. Varicella zoster immunoglobulin (VZIG) – VZIG provides temporary immunity but does not offer long-term protection like the vaccine.
CBIC Infection Control Reference
APIC guidelines recommend placing patients with active shingles in a room with individuals immune to varicella, such as those vaccinated.
An infection preventionist is calculating measures of central tendency regarding duration of a surgical procedure using this data set: 2, 2, 3, 4, and 9. Which of the following statements is correct?
Options:
The median is 2.
The mode is 3.
The mean is 4.
The standard deviation is 7.
Answer:
CExplanation:
Measures of central tendency (mean, median, mode) and dispersion (standard deviation) are statistical tools used to summarize data, such as the duration of surgical procedures, which can help infection preventionists identify trends or risks for surgical site infections. The Certification Board of Infection Control and Epidemiology (CBIC) supports the use of data analysis in the "Surveillance and Epidemiologic Investigation" domain, aligning with epidemiological principles outlined by the Centers for Disease Control and Prevention (CDC). The question provides a data set of 2, 2, 3, 4, and 9, and requires determining the correct statement by calculating these measures.
Mean: The mean is the average of the data set, calculated by summing all values and dividing by the number of observations. For the data set 2, 2, 3, 4, and 9:(2 + 2 + 3 + 4 + 9) ÷ 5 = 20 ÷ 5 = 4. Thus, the mean is 4, making Option C correct.
Median: The median is the middle value when the data set is ordered. With five values (2, 2, 3, 4, 9), the middle value is the third number, which is 3. Option A states the median is 2, which is incorrect.
Mode: The mode is the most frequently occurring value. In this data set, 2 appears twice, while 3, 4, and 9 appear once each, making 2 the mode. Option B states the mode is 3, which is incorrect.
Standard Deviation: The standard deviation measures the spread of data around the mean. For a small data set like this, the calculation involves finding the variance (average of squared differences from the mean) and taking the square root. The mean is 4, so the deviations are: (2-4)² = 4, (2-4)² = 4, (3-4)² = 1, (4-4)² = 0, (9-4)² = 25. The sum of squared deviations is 4 + 4 + 1 + 0 + 25 = 34. The variance is 34 ÷ 5 = 6.8, and the standard deviation is √6.8 ≈ 2.61 (not 7). Option D states the standard deviation is 7, which is incorrect without further context (e.g., a population standard deviation with n-1 denominator would be √34 ≈ 5.83, still not 7).
The CBIC Practice Analysis (2022) and CDC guidelines encourage accurate statistical analysis to inform infection control decisions, such as assessing surgical duration as a risk factor for infections. Based on the calculations, the mean of 4 is the only correct statement among the options, confirming Option C as the answer. Note that the standard deviation of 7 might reflect a miscalculation or misinterpretation (e.g., using a different formula or data set), but with the given data, it does not hold.
Which of the following statements characterizes the proper use of chemical disinfectants?
Options:
All items to be processed must be cleaned prior to being submerged in solution.
The label on the solution being used must indicate that it kills all viable micro-organisms.
The solution should be adaptable for use as an antiseptic.
A chemical indicator must be used with items undergoing high-level disinfection.
Answer:
AExplanation:
The proper use of chemical disinfectants is a critical aspect of infection control, as outlined by the Certification Board of Infection Control and Epidemiology (CBIC). Chemical disinfectants are used to eliminate or reduce pathogenic microorganisms on inanimate objects, and their effective application requires adherence to specific protocols to ensure safety and efficacy. Let’s evaluate each option based on infection control standards:
A. All items to be processed must be cleaned prior to being submerged in solution.: This statement is a fundamental principle of disinfectant use. Cleaning (e.g., removing organic material such as blood, tissue, or dirt) is a prerequisite before disinfection because organic matter can inactivate or reduce the effectiveness of chemical disinfectants. The CBIC emphasizes that proper cleaning is the first step in the disinfection process to ensure that disinfectants can reach and kill microorganisms. This step is universally required for all levels of disinfection (low, intermediate, and high), making it a characterizing feature of proper use.
B. The label on the solution being used must indicate that it kills all viable micro-organisms.: This statement is misleading. No disinfectant can be guaranteed to kill 100% of all viable microorganisms under all conditions, as efficacy depends on factors like contact time, concentration, and the presence of organic material. Disinfectant labels typically indicate the types of microorganisms (e.g., bacteria, viruses, fungi) and the level of disinfection (e.g., high-level, intermediate-level) they are effective against, based on standardized tests (e.g., EPA or FDA guidelines). Claiming that a solution kills all viable microorganisms is unrealistic and not a requirement for proper use; instead, the label must specify the intended use and efficacy, which varies by product.
C. The solution should be adaptable for use as an antiseptic.: An antiseptic is a chemical agent used on living tissue (e.g., skin) to reduce microbial load, whereas a disinfectant is used on inanimate surfaces. While some chemicals (e.g., alcohol) can serve both purposes, this is not a requirement for proper disinfectant use. The adaptability of a solution for antiseptic use is irrelevant to its classification or application as a disinfectant, which focuses on environmental or equipment decontamination. This statement does not characterize proper disinfectant use.
D. A chemical indicator must be used with items undergoing high-level disinfection.: Chemical indicators (e.g., test strips or tapes) are used to verify that the disinfection process has met certain parameters (e.g., concentration or exposure time), particularly in sterilization or high-level disinfection (HLD). While this is a recommended practice for quality assurance in HLD (e.g., with glutaraldehyde or hydrogen peroxide), it is not a universal requirement for all chemical disinfectant use. HLD applies specifically to semi-critical items (e.g., endoscopes), and the need for indicators depends on the protocol and facility standards. This statement is too narrow and specific to characterize the proper use of chemical disinfectants broadly.
The correct answer is A, as cleaning prior to disinfection is a foundational and universally applicable step in the proper use of chemical disinfectants. This aligns with CBIC guidelines, which stress the importance of a clean surface to maximize disinfectant efficacy and prevent infection transmission in healthcare settings.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain IV: Environment of Care, which mandates cleaning as a prerequisite for effective disinfection.
CBIC Examination Content Outline, Domain III: Prevention and Control of Infectious Diseases, which includes protocols for the proper use of disinfectants, emphasizing pre-cleaning.
CDC Guidelines for Disinfection and Sterilization in Healthcare Facilities (2021), which reinforce that cleaning must precede disinfection to ensure efficacy.
The MOST common route of infection for healthcare-associated (HA) pneumonia is:
Options:
Aspiration of bacteria from the oropharynx
Person-to-person spread from healthcare personnel’s (HCP) hands
Contamination from small-volume nebulizers
Droplet inhalation from large-volume humidifiers
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies aspiration of bacteria from the oropharynx as the most common route of infection for healthcare-associated pneumonia, including hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP). In hospitalized patients—especially those who are critically ill, sedated, intubated, or have impaired consciousness—the normal defense mechanisms that prevent aspiration are compromised.
Colonization of the oropharynx with pathogenic organisms occurs rapidly in hospitalized patients due to factors such as antibiotic exposure, underlying illness, poor oral hygiene, and use of invasive devices. Microaspiration of contaminated oral and gastric secretions into the lower respiratory tract is a frequent event and represents the primary mechanism by which pathogens reach the lungs. This risk is significantly increased in patients receiving mechanical ventilation or those positioned supine.
The other options represent less common routes. Transmission from healthcare personnel hands (Option B) contributes indirectly by facilitating colonization but is not the primary route of pneumonia development. Contaminated nebulizers (Option C) and humidifiers (Option D) have been associated with outbreaks but are now uncommon causes due to improved equipment design and maintenance practices.
For CIC® exam preparation, it is essential to recognize that preventive strategies for HA pneumonia focus heavily on reducing aspiration risk, including head-of-bed elevation, oral care protocols, and minimizing sedation—directly addressing the most common route of infection.
Which of the following processes is a critical step in sterile reprocessing of surgical instrumentation?
Options:
Send sterile processing disposable and reusable instrumentation for sorting.
Wrap instruments in disposable pads for protection until transporting to sterile processing.
Hold dirty instruments until the evening shift to minimize handling before returning to sterile processing.
Remove bioburden at the point of care and keep instrumentation damp until it reaches the sterile processing department.
Answer:
DExplanation:
A critical principle emphasized in the Certification Study Guide (6th edition) is that instrument reprocessing begins at the point of use, not in the sterile processing department. Immediate removal of gross soil and organic material—referred to as bioburden—prevents drying of blood, tissue, and other debris on instruments. Dried organic material significantly interferes with subsequent cleaning, disinfection, and sterilization processes, reducing the effectiveness of these steps and increasing the risk of surgical site infections.
The study guide explains that instruments should be kept moist or damp after use, typically by using an approved enzymatic spray, damp towel, or transport container, to prevent soil from adhering firmly to surfaces and lumens. This practice protects both the integrity of the instruments and the safety of personnel handling them. Delaying cleaning or allowing instruments to dry increases microbial load and biofilm formation, which are difficult to remove during later processing stages.
The incorrect options conflict with infection prevention standards: holding dirty instruments increases contamination risk; wrapping instruments in pads does not address bioburden; and sending instruments for sorting without point-of-care decontamination violates best practices. Proper point-of-care treatment is foundational to safe, effective sterile processing and is consistently tested on the CIC exam.
Which of the following is NOT valuable when using a graph to display data?
Options:
Title with topic of data
Facility and time frame for data
Published trends for data comparison
X and Y axes labels and annotation where indicated
Answer:
CExplanation:
The Certification Study Guide (6th edition) explains that graphs are most effective when they clearly communicate who, what, when, and how regarding the data being presented. Essential elements include a descriptive title, identification of the facility and time frame, and properly labeled X and Y axes with annotations as needed. These components ensure that the viewer can accurately interpret the data without additional explanation.
Published trends for data comparison, while potentially useful in separate analyses or reports, are not required elements of an individual graph and do not inherently improve the clarity of data display. Including external published trends can actually confuse interpretation if definitions, populations, or surveillance methodologies differ from the local data being presented. The study guide cautions against mixing datasets with different assumptions or collection methods in a single visual display unless clearly contextualized.
Titles clarify the subject of the graph, facility and time frame provide essential context, and axis labels ensure the viewer understands what is being measured. These are foundational principles of data visualization emphasized in infection prevention reporting and communication.
CIC exam questions frequently test the ability to distinguish between essential graph components and supplementary analytical tools. Recognizing that published comparison trends are not required—and may be misleading—reinforces good data communication practices and supports accurate interpretation by leadership and frontline staff.
The infection preventionist (IP) is notified about the recall of certain ice machines because of a faulty filtration device. These ice machines are located on several units throughout the facility. What is the BEST action the IP should take?
Options:
Culture all ice machines for Legionella
Report a waterborne disease outbreak to Administration
Identify all patients who have been served ice from these machines
Supply an alternative source of ice while investigating further
Answer:
DExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that when a device recall involves potential contamination risk, the infection preventionist’s first priority is risk mitigation and prevention of further exposure. In this scenario, the recall of ice machines due to a faulty filtration device represents a potential waterborne contamination risk, even in the absence of confirmed infections.
The best immediate action is to remove the recalled ice machines from service and provide an alternative source of ice while further investigation and corrective actions are underway. This step promptly eliminates the exposure pathway and protects patients, staff, and visitors from possible contamination. The Study Guide stresses that interruption of use is the most effective initial control measure when equipment safety is in question.
Option A is incorrect because culturing ice machines is not the first step and is not routinely recommended without clinical indication. Option B is inappropriate because there is no evidence of a confirmed outbreak. Option C may be necessary later if exposure investigation becomes warranted, but it should not precede immediate risk control.
For the CIC® exam, it is essential to recognize that eliminating exposure takes precedence over testing or notification activities. Supplying an alternative ice source while investigating further aligns with risk management principles, patient safety priorities, and evidence-based infection prevention practice.
==========
What factors contribute to the development of bacteria-containing biofilms within channels of endoscopes?
Options:
Frequent use of enzymatic detergents.
Inadequate cleaning and reprocessing protocols.
Use of chlorine- and monochloramine-based products.
Using a central reprocessing area rather than a local reprocessing area.
Answer:
BExplanation:
The Certification Study Guide (6th edition) identifies inadequate cleaning and reprocessing protocols as the primary factor contributing to the formation of bacteria-containing biofilms within endoscope channels. Endoscopes have long, narrow lumens and complex internal surfaces that are particularly vulnerable to biofilm formation when organic material is not thoroughly removed. Biofilms develop when microorganisms adhere to surfaces and become embedded within a protective extracellular matrix, which significantly reduces the effectiveness of disinfectants and sterilants.
The study guide emphasizes that cleaning is the most critical step in endoscope reprocessing. Failure to promptly and thoroughly clean channels—such as delayed cleaning, insufficient brushing, inadequate flushing, or improper detergent use—allows organic debris and moisture to remain, creating ideal conditions for microbial attachment and biofilm development. Once established, biofilms are difficult to eliminate and have been implicated in healthcare-associated infections linked to endoscopic procedures.
The incorrect options describe practices that do not promote biofilm formation. Enzymatic detergents, when used correctly, support removal of organic material. Chlorine-based products are not standard for endoscope channel reprocessing and are not the primary cause of biofilm development. Centralized reprocessing areas are considered best practice because they support standardized procedures, trained personnel, and quality control.
This concept is frequently tested on the CIC exam, reinforcing that breakdowns in basic cleaning and reprocessing practices pose the greatest risk for biofilm formation and patient harm.
A hospital is experiencing an increase in multidrug-resistant Acinetobacter baumannii infections in the intensive care unit (ICU). The infection preventionist's FIRST action should be to:
Options:
Implement universal contact precautions for all ICU patients.
Conduct an epidemiologic investigation to identify potential sources.
Perform environmental sampling to detect Acinetobacter on surfaces.
Initiate decolonization protocols for all ICU patients.
Answer:
BExplanation:
Epidemiologic Investigation:
The first step in an outbreak response is to characterize cases by person, place, and time.
Identifying common exposures (e.g., ventilators, catheters, or contaminated surfaces) helps determine the source.
Why Other Options Are Incorrect:
A. Universal contact precautions: Premature; precautions should be tailored based on transmission patterns.
C. Environmental sampling: Should be done after identifying epidemiologic links.
D. Decolonization protocols: Not routinely recommended for Acinetobacter outbreaks.
CBIC Infection Control References:
CIC Study Guide, "Epidemiologic Investigations in Outbreaks," Chapter 4.
A 2-yoar-old girl is admitted with a fractured tibia. At birth, she was diagnosed with congenital cytomegalovirus (CMV). Which of the following barrier precautions is appropriate for healthcare personnel caring for her?
Options:
Wear masks and gloves
Wear gloves when handling body fluids
No barrier precautions are needed
Use gowns, masks, gloves, and a private room
Answer:
BExplanation:
Standard Precautions are sufficient for congenital cytomegalovirus (CMV), which means that gloves should be used when handling body fluids. CMV is primarily transmitted via direct contact with saliva, urine, or blood.
Why the Other Options Are Incorrect?
A. Wear masks and gloves – Masks are not necessary unless performing high-risk aerosol-generating procedures.
C. No barrier precautions are needed – Gloves are required when handling bodily fluids to prevent transmission.
D. Use gowns, masks, gloves, and a private room – CMV does not require Contact or Airborne Precautions.
CBIC Infection Control Reference
APIC guidelines state that CMV transmission is prevented using Standard Precautions, primarily with glove use for body fluid contact.
There is an influenza epidemic in a community. To prevent transmission of influenza in a facility, the MOST rapidly effective measure an infection preventionist should recommend is to:
Options:
Use droplet precautions empirically for all residents suspected to have influenza.
Immediately immunize patient care staff.
Immunize patients and patient care staff.
Notify the local health department.
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that when influenza is circulating in the community, early recognition and immediate implementation of infection control measures are essential to prevent transmission within healthcare facilities. Among available interventions, the most rapidly effective measure is the empiric use of Droplet Precautions for patients suspected of having influenza.
Influenza is transmitted primarily through respiratory droplets generated by coughing, sneezing, or talking. Patients may be infectious before laboratory confirmation is available, making empiric precautions critical. Initiating Droplet Precautions—such as use of surgical masks, patient placement considerations, and limiting movement—immediately reduces the risk of person-to-person transmission and protects healthcare personnel and other patients.
While vaccination of staff and patients (Options B and C) is an essential long-term prevention strategy, it does not provide immediate protection because immunity develops over days to weeks. Therefore, vaccination alone is not the most rapidly effective intervention during an active outbreak. Option D, notifying the local health department, is important for surveillance and public health coordination but does not directly and immediately reduce transmission within the facility.
For CIC® exam preparation, it is crucial to distinguish between immediate containment measures and longer-term prevention strategies. Empiric Droplet Precautions for suspected influenza cases represent the fastest and most effective method to interrupt transmission during an influenza epidemic.
What method of evaluation will BEST identify a staff member’s competency with reprocessing medical devices?
Options:
Verbalize the importance of reprocessing.
Demonstrate the appropriate sterilization procedure.
Describe the facility’s sterilization policies and procedures.
Obtain a score of 100% on a post-test following a reprocessing course.
Answer:
BExplanation:
The correct answer is B, "Demonstrate the appropriate sterilization procedure," as this method of evaluation will best identify a staff member’s competency with reprocessing medical devices. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, competency in reprocessing medical devices—such as cleaning, disinfection, and sterilization—requires not only theoretical knowledge but also the practical ability to perform the tasks correctly and safely. Demonstration allows the infection preventionist (IP) to directly observe the staff member’s hands-on skills, adherence to protocols (e.g., AAMI ST79), and ability to handle equipment, ensuring that the reprocessing process effectively prevents healthcare-associated infections (HAIs) (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.3 - Assess competence of healthcare personnel). This method provides tangible evidence of proficiency, as it tests the application of knowledge in a real or simulated setting, which is critical for ensuring patient safety.
Option A (verbalize the importance of reprocessing) assesses understanding and awareness, but it is a theoretical exercise that does not confirm the ability to perform the task, making it insufficient for evaluating competency. Option C (describe the facility’s sterilization policies and procedures) tests knowledge of guidelines, which is a component of competence but lacks the practical demonstration needed to verify skill execution. Option D (obtain a score of 100% on a post-test following a reprocessing course) measures theoretical knowledge and retention, but a perfect score does not guarantee practical ability, as it does not assess hands-on performance or problem-solving under real conditions.
The focus on demonstration aligns with CBIC’s emphasis on assessing competence through observable performance, ensuring that staff can reliably reprocess devices to maintain a sterile environment (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). This method supports a comprehensive evaluation, aligning with best practices for training and competency assessment in healthcare settings.
A hospital experiencing an increase in catheter-associated urinary tract infections (CAUTI) implements a quality improvement initiative. Which of the following interventions is MOST effective in reducing CAUTI rates?
Options:
Routine urine cultures for all catheterized patients every 48 hours.
Implementing nurse-driven protocols for early catheter removal.
Replacing indwelling urinary catheters with condom catheters for all male patients.
Using antibiotic-coated catheters in all ICU patients.
Answer:
BExplanation:
Nurse-driven catheter removal protocols have been shown to significantly reduce CAUTI rates by minimizing unnecessary catheter use.
Routine urine cultures (A) lead to overtreatment of asymptomatic bacteriuria.
Condom catheters (C) are helpful in certain cases but are not universally effective.
Antibiotic-coated catheters (D) have mixed evidence regarding their effectiveness.
CBIC Infection Control References:
APIC Text, "CAUTI Prevention Strategies," Chapter 10.
When a Staphylococcus aureus outbreak is suspected, cultures of which of the following sites will MOST likely identify carriers?
Options:
Nose
Hands
Throat
Rectum
Answer:
AExplanation:
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the anterior nares (nose) as the most common and reliable site for colonization with Staphylococcus aureus, including methicillin-resistant Staphylococcus aureus (MRSA). During suspected outbreaks, culturing the nares is the most effective method for identifying persistent carriers, particularly among healthcare personnel or patients who may serve as reservoirs for transmission.
Nasal carriage of S. aureus is well established in epidemiologic literature and infection prevention practice. Individuals may be persistent carriers, intermittent carriers, or non-carriers, with persistent nasal carriers posing the highest risk for transmission and subsequent infection. The Study Guide emphasizes that nasal colonization strongly correlates with both endogenous infection risk and spread to others, making it the preferred screening site during outbreak investigations.
Hands (Option B) may transiently harbor S. aureus, but hand contamination is temporary and highly variable, making it less useful for identifying long-term carriers. Throat (Option C) and rectum (Option D) are not primary colonization sites for S. aureus and are not routinely used in outbreak screening unless specifically indicated by epidemiologic data.
For CIC® exam purposes, this question reinforces a core infection prevention principle: the anterior nares are the primary reservoir for Staphylococcus aureus, and nasal cultures are the most effective method for identifying carriers during outbreak investigations.
The infection preventionist understands that the heating, ventilation and air conditioning (HVAC) systems in the facility can be a risk factor for healthcare-acquired infections. What is the MOST likely risk from the HVAC system for patients in a Pediatric Oncology unit?
Options:
Methicillin-resistant Staphylococcus aureus (MRSA)
Norovirus
Aspergillus spp.
Clostridioides difficile
Answer:
CExplanation:
Patients in pediatric oncology units are highly immunocompromised, making them particularly susceptible to opportunistic fungal infections such as Aspergillus spp. HVAC systems, especially if improperly maintained or contaminated, can disseminate fungal spores into patient care areas.
According to the APIC Text (Chapter 116 – HVAC Systems), fungal spores such as Aspergillus can be transmitted via HVAC systems. These infections have been linked to contaminated air ducts, faulty air filters, and construction-related air disturbances. Outbreaks of aspergillosis are frequently associated with construction near patient care areas and are particularly dangerous for immunocompromised patients, including pediatric oncology patients.
Additional data from APIC Text (Chapter 45 – Infection Prevention in Oncology Patients) reinforces that Aspergillus spp. infections in oncology and immunocompromised patients are primarily airborne and are most often disseminated via HVAC systems.
Incorrect answer rationale:
A. MRSA – Typically spread via direct contact, not HVAC.
B. Norovirus – Spread via fecal-oral route and contaminated surfaces, not airborne HVAC.
D. Clostridioides difficile – Spread via contact with spores on surfaces, not through the air.
Unlock CIC Features
- CIC All Real Exam Questions
- CIC Exam easy to use and print PDF format
- Download Free CIC Demo (Try before Buy)
- Free Frequent Updates
- 100% Passing Guarantee by Activedumpsnet
Questions & Answers PDF Demo
- CIC All Real Exam Questions
- CIC Exam easy to use and print PDF format
- Download Free CIC Demo (Try before Buy)
- Free Frequent Updates
- 100% Passing Guarantee by Activedumpsnet
